The catalytic asymmetric polyene cyclization of homofarnesol to ambrox
IF 50.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
Abstract
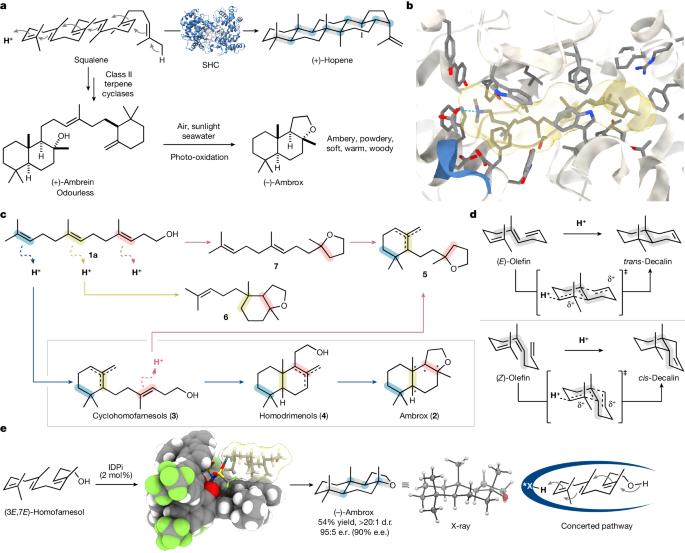
Polyene cyclizations are among the most complex and challenging transformations in biology. In a single reaction step, multiple carbon–carbon bonds, ring systems and stereogenic centres are constituted from simple, acyclic precursors1–3. Simultaneously achieving this kind of precise control over product distribution and stereochemistry poses a formidable task for chemists. In particular, the polyene cyclization of (3E,7E)-homofarnesol to the valuable naturally occurring ambergris odorant (−)-ambrox is recognized as a longstanding challenge in chemical synthesis1,4–7. Here we report a diastereoselective and enantioselective synthesis of (−)-ambrox and the sesquiterpene lactone natural product (+)-sclareolide by a catalytic asymmetric polyene cyclization by using a highly Brønsted-acidic and confined imidodiphosphorimidate catalyst in the presence of fluorinated alcohols. Several experiments, including deuterium-labelling studies, suggest that the reaction predominantly proceeds through a concerted pathway in line with the Stork–Eschenmoser hypothesis8–10. Mechanistic studies show the importance of the enzyme-like microenvironment of the imidodiphosphorimidate catalyst for attaining exceptionally high selectivities, previously thought to be achievable only in enzyme-catalysed polyene cyclizations. The catalytic asymmetric polyene cyclization of homofarnesol to ambrox is achieved using a highly Brønsted-acidic and confined imidodiphosphorimidate catalyst.
催化均苯四甲酚不对称多烯环化为氨溴索。
多烯环化是生物学中最复杂、最具挑战性的转化过程之一。在一个反应步骤中,多个碳-碳键、环系统和立体中心从简单的无环前体中形成1-3。同时实现对产物分布和立体化学的这种精确控制对化学家来说是一项艰巨的任务。其中,将 (3E,7E)-homofarnesol 的多烯环化反应转化为珍贵的天然龙涎香气味剂 (-)-ambrox 是化学合成领域的一项长期挑战1,4-7。在此,我们报告了一种非对映选择性合成 (-)-ambrox 和倍半萜内酯天然产物 (+)-sclareolide 的方法,该方法是在氟化醇存在下,使用一种高度布氏酸性和封闭的亚胺二磷酰亚胺催化剂,通过催化不对称多烯环化反应合成 (-)-ambrox 和倍半萜内酯天然产物 (+)-sclareolide。包括氘标记研究在内的多项实验表明,该反应主要通过符合斯托克-埃申莫瑟假说(Stork-Eschenmoser hypothesis)的协同途径进行8-10。机理研究表明,亚胺二磷酰亚胺催化剂的酶样微环境对获得极高的选择性非常重要,而以前认为只有在酶催化的多烯环化反应中才能实现这种选择性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


