Amination of Phenols and Halophenols via Pyridinium–Iridium Dual Photocatalysis
IF 13.1
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
In this study, we present a photochemical method for the amination of phenols (C–H) and halophenols (SNAr), facilitated by dual catalytic pathways involving both Ir(III) photocatalysis and phenol–pyridinium electron donor–acceptor complexation. By incorporating a pyridinium additive, we achieved efficient C–N coupling between phenols and diverse aromatic nitrogen nucleophiles, delivering high yields (up to 99%) across a wide range of substrates, including pharmaceuticals and natural products. We investigate reaction selectivity and substrate compatibility/limitations through a combination of experimental and computational techniques. Moreover, we highlight the synthetic versatility of the amination products through various late-stage functionalizations including the grafting of two different heteroarenes onto one phenol scaffold.
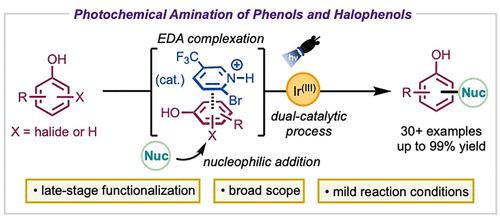
通过吡啶-铱双光催化技术胺化苯酚和卤苯酚
在本研究中,我们提出了一种用于苯酚(C-H)和卤代苯酚(SNAr)胺化反应的光化学方法,该方法通过涉及 Ir(III) 光催化和苯酚-吡啶电子供体-受体络合的双重催化途径来实现。通过加入吡啶添加剂,我们实现了苯酚与多种芳香族氮亲核物之间的高效 C-N 偶联,在包括药物和天然产物在内的多种底物中实现了高产率(高达 99%)。我们结合实验和计算技术研究了反应选择性和底物兼容性/限制。此外,我们还通过各种后期官能化(包括在一个苯酚支架上接枝两种不同的杂环戊烯),强调了胺化产物的合成多功能性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


