Manipulating Superexchange Interaction of Ru–O–Fe Sites for Enhanced Electrocatalytic Nitrate-to-Ammonia Selectivity
IF 11.3
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
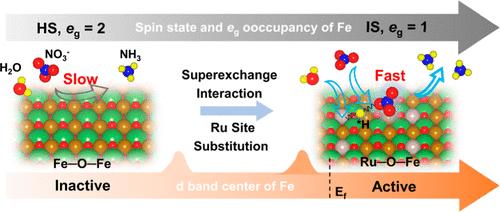
Fe-based catalysts are promising for electrochemical nitrate reduction, but their selectivity is limited by the multielectron/proton transfer reaction steps. Here, we propose optimizing the eg-orbital electron occupancy by regulating the superexchange interaction of the Fe site to improve the NH3 production performance. Our experimental and theoretical prediction results confirmed that Ru–O–Fe sites in double perovskite iron oxides (LaFe0.9Ru0.1O3) have more significant superexchange interactions, mainly manifested by O-anion-mediated electron transfer from Ru to Fe cations. Ru alters Fe’s spin configuration through Ru–O–Fe orbital hybridization, transitioning from a high-spin (HS, eg ≈ 2) to an intermediate-spin state (eg ≈ 1). This transition promotes NO3– adsorption and lowers the hydrogenation energy barrier of the *NO intermediate. Consequently, LaFe0.9Ru0.1O3 could efficiently convert NO3– to NH3, achieving rates of 0.75 mmol·h–1·cm–2 with a Faraday efficiency of 98.5%. Remarkably, the NH3 selectivity was as high as 90.7%, which represents almost the best catalyst to date.
操纵 Ru-O-Fe 位点的超交换相互作用以增强硝酸-氨的电催化选择性
铁基催化剂有望用于电化学硝酸盐还原,但其选择性受到多电子/质子转移反应步骤的限制。在此,我们建议通过调节 Fe 位点的超交换相互作用来优化电子轨道的电子占有率,从而提高 NH3 生成性能。我们的实验和理论预测结果证实,双包晶铁氧化物(LaFe0.9Ru0.1O3)中的Ru-O-Fe位点具有更显著的超交换相互作用,主要表现为O-阴离子介导的电子从Ru转移到Fe阳离子。Ru 通过 Ru-O-Fe 轨道杂化改变 Fe 的自旋构型,从高自旋(HS,例如 ≈ 2)过渡到中自旋状态(例如 ≈ 1)。这种转变促进了 NO3- 的吸附,并降低了 *NO 中间体的氢化能垒。因此,LaFe0.9Ru0.1O3 能有效地将 NO3- 转化为 NH3,转化率达到 0.75 mmol-h-1-cm-2,法拉第效率为 98.5%。值得注意的是,NH3 的选择性高达 90.7%,几乎是迄今为止最好的催化剂。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


