Synthesis and Structural Characterization of Lithium Ionic Conductors Li2MS3 (M = Si, Si0.5Ge0.5, Ge) and Li16Ge5S18
IF 7.2
2区 材料科学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
To develop inorganic Li conductors for all-solid-state Li-ion batteries, the present study clarified temperature-dependent structural changes in the Li2MS3 (M = Si, Si0.5Ge0.5, Ge) phases, where new crystal structures favoring ionic conduction are potentially formed. Structural analysis was performed on powder X-ray diffraction data obtained at high temperatures. At 600 °C, tetragonal phases were formed in all three cases. Meanwhile, structural changes on cooling from 600 °C were different according to the M species, leading to various crystal systems at room temperature (monoclinic for M = Si, orthorhombic for Si0.5Ge0.5, and hexagonal for Ge). Their ionic conductivities at 25 °C were 2.3 × 10–5, 1.7 × 10–7, and 8.6 × 10–9 S cm–1, respectively, indicating that the conductivity depends significantly on the framework of the crystal structure. These phase identification results were further utilized to search the Li2S–GeS2 pseudobinary system, revealing that the structure of Li16Ge5S18 could not be assigned to any polymorphs identified for Li2MS3. This new crystalline phase showed an ionic conductivity of 7.2 × 10–5 S cm–1 at room temperature, which is 4 orders of magnitude higher than that of hexagonal Li2GeS3. This study highlights a drastic increase in the lithium conductivity achieved by controlling the crystal structure of the Li2MS3-related materials.
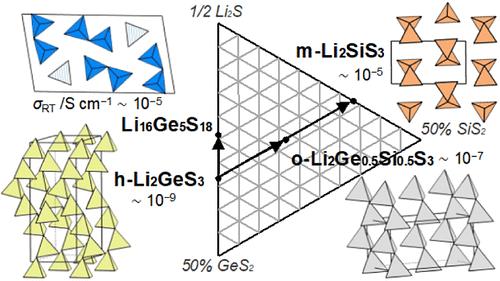
锂离子导体 Li2MS3(M = Si、Si0.5Ge0.5、Ge)和 Li16Ge5S18 的合成与结构表征
为了开发用于全固态锂离子电池的无机锂导体,本研究阐明了 Li2MS3(M = Si、Si0.5Ge0.5、Ge)相随温度变化而发生的结构变化,其中可能形成了有利于离子传导的新晶体结构。对高温下获得的粉末 X 射线衍射数据进行了结构分析。在 600 °C 时,三种情况下都形成了四方相。同时,从 600 ℃ 冷却时的结构变化因 M 种类的不同而不同,导致室温下形成各种晶系(M = Si 时为单斜晶系,Si0.5Ge0.5 时为正长晶系,Ge 时为六方晶系)。它们在 25 °C 时的离子电导率分别为 2.3 × 10-5、1.7 × 10-7 和 8.6 × 10-9 S cm-1,表明电导率在很大程度上取决于晶体结构的框架。这些相鉴定结果被进一步用于搜索 Li2S-GeS2 伪二元体系,结果发现 Li16Ge5S18 的结构无法归属于为 Li2MS3 鉴定的任何多晶体。这种新晶相在室温下的离子电导率为 7.2 × 10-5 S cm-1,比六方 Li2GeS3 的离子电导率高出 4 个数量级。这项研究表明,通过控制 Li2MS3 相关材料的晶体结构,锂电导率得到了大幅提高。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemistry of Materials
工程技术-材料科学:综合
CiteScore
14.10
自引率
5.80%
发文量
929
审稿时长
1.5 months
期刊介绍:
The journal Chemistry of Materials focuses on publishing original research at the intersection of materials science and chemistry. The studies published in the journal involve chemistry as a prominent component and explore topics such as the design, synthesis, characterization, processing, understanding, and application of functional or potentially functional materials. The journal covers various areas of interest, including inorganic and organic solid-state chemistry, nanomaterials, biomaterials, thin films and polymers, and composite/hybrid materials. The journal particularly seeks papers that highlight the creation or development of innovative materials with novel optical, electrical, magnetic, catalytic, or mechanical properties. It is essential that manuscripts on these topics have a primary focus on the chemistry of materials and represent a significant advancement compared to prior research. Before external reviews are sought, submitted manuscripts undergo a review process by a minimum of two editors to ensure their appropriateness for the journal and the presence of sufficient evidence of a significant advance that will be of broad interest to the materials chemistry community.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


