Rhenium-catalyzed synthetic method of indanes and indenes through the C–C bond cleavage of 1,3-dicarbonyl compounds
IF 1.5
4区 化学
Q3 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
When α-alkyl substituted 1,3-dicarbonyl compounds were reacted with alkenes and alkynes in the presence of a rhenium catalyst, the reaction proceeded through the elimination of the 1,3-dicarbonyl unit to form the corresponding indanes and indenes in moderate to good yields, respectively.
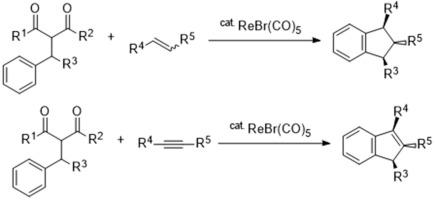
通过 1,3 二羰基化合物的 C-C 键裂解合成茚和茚烯的铼催化方法
当α-烷基取代的 1,3-二羰基化合物在铼催化剂存在下与烯烃和炔烃反应时,反应通过消除 1,3-二羰基单元进行,生成相应的茚和茚烯(产率分别为中等和良好)。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Tetrahedron Letters
化学-有机化学
CiteScore
3.50
自引率
5.60%
发文量
521
审稿时长
28 days
期刊介绍:
Tetrahedron Letters provides maximum dissemination of outstanding developments in organic chemistry. The journal is published weekly and covers developments in techniques, structures, methods and conclusions in experimental and theoretical organic chemistry. Rapid publication of timely and significant research results enables researchers from all over the world to transmit quickly their new contributions to large, international audiences.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


