Catalytic enantioselective synthesis of hydroxyimino tetrahydrobenzofuranones bearing trifluoromethylated quaternary stereocenter via tandem Michael addition/interrupted Nef reactions
IF 1.5
4区 化学
Q3 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
Tetrahydrobenzofuran derivatives are widely distributed in biologically active molecules and natural products. Herein, we reported an asymmetric tandem Michael addition/interrupted Nef reaction triggered by nitromethane directly as Michael donor catalyzed by chiral 2-aminobenzimidazole bifunctional organocatalyst. A series of enantiomerically enriched trifluoromethylated hydroxyimino tetrahydrobenzofuranones were synthesized in moderate to good isolated yields (up to 97 %) and enantioselectivities (up to 80 % ee).
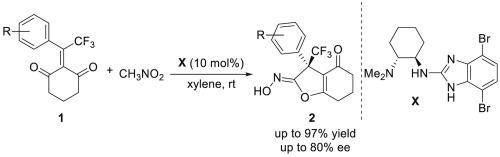
通过串联迈克尔加成/间歇奈夫反应催化对映体选择性合成带有三氟甲基季立体中心的羟基亚氨基四氢苯并呋喃酮
四氢苯并呋喃衍生物广泛分布于生物活性分子和天然产物中。在此,我们报道了在手性 2-aminobenzimidazole 双功能有机催化剂的催化下,由硝基甲烷直接作为迈克尔供体引发的不对称串联迈克尔加成/间断 Nef 反应。合成了一系列对映体富集的三氟甲基化羟基亚氨基四氢苯并呋喃酮,分离产率(高达 97%)和对映体选择性(高达 80%ee)均为中等至良好。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Tetrahedron Letters
化学-有机化学
CiteScore
3.50
自引率
5.60%
发文量
521
审稿时长
28 days
期刊介绍:
Tetrahedron Letters provides maximum dissemination of outstanding developments in organic chemistry. The journal is published weekly and covers developments in techniques, structures, methods and conclusions in experimental and theoretical organic chemistry. Rapid publication of timely and significant research results enables researchers from all over the world to transmit quickly their new contributions to large, international audiences.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


