Cytotoxic T cells drive doxorubicin-induced cardiac fibrosis and systolic dysfunction
IF 9.4
Q1 CARDIAC & CARDIOVASCULAR SYSTEMS
引用次数: 0
Abstract
Doxorubicin, the most prescribed chemotherapeutic drug, causes dose-dependent cardiotoxicity and heart failure. However, our understanding of the immune response elicited by doxorubicin is limited. Here we show that an aberrant CD8+ T cell immune response following doxorubicin-induced cardiac injury drives adverse remodeling and cardiomyopathy. Doxorubicin treatment in non-tumor-bearing mice increased circulating and cardiac IFNγ+CD8+ T cells and activated effector CD8+ T cells in lymphoid tissues. Moreover, doxorubicin promoted cardiac CD8+ T cell infiltration and depletion of CD8+ T cells in doxorubicin-treated mice decreased cardiac fibrosis and improved systolic function. Doxorubicin treatment induced ICAM-1 expression by cardiac fibroblasts resulting in enhanced CD8+ T cell adhesion and transformation, contact-dependent CD8+ degranulation and release of granzyme B. Canine lymphoma patients and human patients with hematopoietic malignancies showed increased circulating CD8+ T cells after doxorubicin treatment. In human cancer patients, T cells expressed IFNγ and CXCR3, and plasma levels of the CXCR3 ligands CXCL9 and CXCL10 correlated with decreased systolic function. Bayer et al. show that CD8+ T cells contribute to the adverse cardiac effect of doxorubicin administration, promoting fibroblast activation and inflammation through a mechanism dependent on IFNγ and the degranulation of granzyme B.
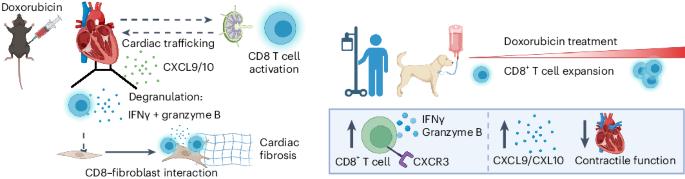
细胞毒性 T 细胞驱动多柔比星诱导的心脏纤维化和收缩功能障碍
多柔比星是最常用的化疗药物,会引起剂量依赖性心脏毒性和心力衰竭。然而,我们对多柔比星引起的免疫反应了解有限。在这里,我们发现多柔比星诱发心脏损伤后,CD8+ T 细胞免疫反应异常会导致不良重塑和心肌病。对非肿瘤小鼠进行多柔比星治疗会增加循环和心脏中的 IFNγ+CD8+ T 细胞,并激活淋巴组织中的效应 CD8+ T 细胞。此外,多柔比星可促进心脏 CD8+ T 细胞浸润,多柔比星治疗小鼠的 CD8+ T 细胞耗竭可减少心脏纤维化并改善收缩功能。多柔比星治疗可诱导心脏成纤维细胞表达 ICAM-1,从而增强 CD8+ T 细胞的粘附和转化、接触依赖性 CD8+ 脱颗粒和释放颗粒酶 B。犬淋巴瘤患者和人类造血恶性肿瘤患者在接受多柔比星治疗后,循环 CD8+ T 细胞增多。在人类癌症患者中,T 细胞表达 IFNγ 和 CXCR3,血浆中 CXCR3 配体 CXCL9 和 CXCL10 的水平与收缩功能下降相关。Bayer 等人的研究表明,CD8+ T 细胞通过一种依赖于 IFNγ 和颗粒酶 B 的脱颗粒机制促进成纤维细胞活化和炎症反应,从而导致了多柔比星给药对心脏的不良影响。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


