Modified Chitosan for Highly Efficient Non‐Invasive Transdermal Delivery of Catalase to Repair and Prevent Skin Photodamages
IF 18.5
1区 材料科学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
Oxidative stress induced by excess reactive oxygen species (ROS) plays a significant role in the onset and progression of numerous skin disorders, necessitating effective antioxidant defenses to prevent and repair oxidative damage. However, existing systemic antioxidant therapies for skin diseases often fall short in efficiently delivering antioxidants to the diseased lesions. In this study, a novel non‐invasive transdermal delivery platform utilizing chitosan grafted with salcaprozate sodium (SCS) is designed for highly efficient delivery of biomolecular enzymes, such as catalase (CAT). After self‐assembling with catalase, the obtained SCS‐CAT nanocomplexes if topically applied in a cream demonstrate highly efficient skin penetration and accumulation. Owing to the ability of CAT to effectively scavenge ROS, topically applied SCS‐CAT nanocomplexes enable remarkable repair and protection effects against ultraviolet radiation B (UVB)‐induced skin photodamages by inhibiting cell apoptosis and inflammation. Moreover, such SCS‐CAT delivery platform holds promise for long‐term skin care applications due to its great biocompatibility. This research presents a simple yet transformative platform for the intradermal delivery of biological enzymes, presenting a promising avenue for treating various inflammatory skin disorders.
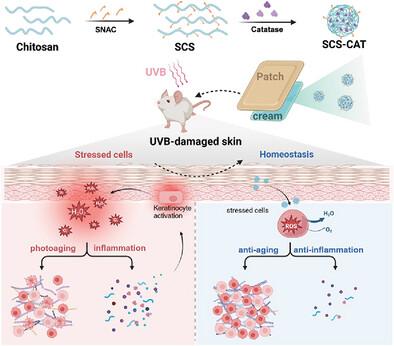
改性壳聚糖用于高效非侵入性经皮输送过氧化氢酶,以修复和预防皮肤光损伤
过量活性氧(ROS)引起的氧化应激在多种皮肤病的发病和发展过程中起着重要作用,因此需要有效的抗氧化防御措施来预防和修复氧化损伤。然而,现有的系统性皮肤病抗氧化疗法往往不能有效地将抗氧化剂输送到病变部位。本研究设计了一种新型的非侵入性透皮给药平台,利用壳聚糖接枝盐己酸钠(SCS)高效给药生物分子酶,如过氧化氢酶(CAT)。在与过氧化氢酶自组装后,获得的 SCS-CAT 纳米复合物在乳霜中局部涂抹后,会显示出高效的皮肤渗透和蓄积能力。由于 CAT 能够有效清除 ROS,外用 SCS-CAT 纳米复合物可通过抑制细胞凋亡和炎症,对紫外线辐射 B(UVB)引起的皮肤光损伤起到显著的修复和保护作用。此外,这种 SCS-CAT 递送平台具有良好的生物相容性,有望用于长期皮肤护理。这项研究为皮内输送生物酶提供了一个简单但具有变革性的平台,为治疗各种炎症性皮肤病提供了一条前景广阔的途径。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Advanced Functional Materials
工程技术-材料科学:综合
CiteScore
29.50
自引率
4.20%
发文量
2086
审稿时长
2.1 months
期刊介绍:
Firmly established as a top-tier materials science journal, Advanced Functional Materials reports breakthrough research in all aspects of materials science, including nanotechnology, chemistry, physics, and biology every week.
Advanced Functional Materials is known for its rapid and fair peer review, quality content, and high impact, making it the first choice of the international materials science community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


