Experimental Study on Vapor–Liquid and Solid–Liquid Equilibria Data for the Regeneration of Biobased Solvents Guaiacol and 2,2,5,5-Tetramethyl Oxolane in Biorefinery Processes
IF 2
3区 工程技术
Q3 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
Solvent regeneration is crucial after liquid–liquid extraction (LLX). This study investigates the vapor–liquid equilibria (VLE) and solid–liquid equilibria (SLE) for regenerating guaiacol and 2,2,5,5-tetramethyl oxolane (TMO) following LLX. The deep eutectic solvent (DES) composed of lactic acid and choline chloride was regenerated by LLX with these biobased solvents after biomass delignification. Both crystallization and evaporation methods have been considered for solvent regeneration. The experimental SLE study in the guaiacol-lactic acid system reveals a solid solution formation, with significant nonideality in the liquidus and solidus lines at guaiacol weight fractions around 0.80 to 1. A two-step crystallization process is conceptualized, concentrating guaiacol in the solid state, obtaining noticeable reductions in HMF and furfural concentrations in the solid phase, with guaiacol yields of 57.0% in the first stage and 39.8% in the second stage. Additionally, vapor pressures of guaiacol (400.59–439.67 K) and TMO (318.43–372.16 K) have been measured to facilitate solvent regeneration simulations via evaporation or distillation. Antoine parameters were fitted for both solvents to experimental vapor pressure data, with an average deviation of 0.05 K for guaiacol and 0.07 K for TMO.
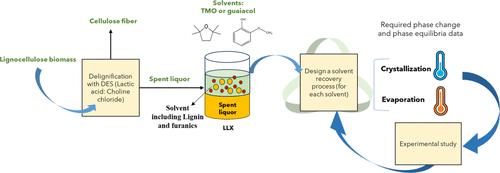
生物炼制工艺中再生生物基溶剂愈创木酚和 2,2,5,5- 四甲基羰基酚的气液和固液平衡数据实验研究
液液萃取(LLX)后的溶剂再生至关重要。本研究探讨了液-液萃取再生愈创木酚和 2,2,5,5- 四甲基氧杂环戊烷(TMO)时的汽液平衡(VLE)和固液平衡(SLE)。乳酸和氯化胆碱组成的深共晶溶剂(DES)在生物质脱木质后通过 LLX 与这些生物基溶剂再生。结晶和蒸发两种方法都被用于溶剂再生。在愈创木酚-乳酸体系中进行的 SLE 实验研究表明,在愈创木酚重量分数约为 0.80 至 1 时,固溶体形成,液相线和固相线具有显著的非线性。我们设想了一个两步结晶过程,在固态下浓缩愈创木酚,从而显著降低固相中 HMF 和糠醛的浓度,第一阶段愈创木酚的产量为 57.0%,第二阶段为 39.8%。此外,还测量了愈创木酚(400.59-439.67 K)和 TMO(318.43-372.16 K)的蒸汽压,以便通过蒸发或蒸馏进行溶剂再生模拟。两种溶剂的安托万参数都与实验蒸汽压力数据相匹配,愈创木酚和 TMO 的平均偏差分别为 0.05 K 和 0.07 K。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Chemical & Engineering Data
工程技术-工程:化工
CiteScore
5.20
自引率
19.20%
发文量
324
审稿时长
2.2 months
期刊介绍:
The Journal of Chemical & Engineering Data is a monthly journal devoted to the publication of data obtained from both experiment and computation, which are viewed as complementary. It is the only American Chemical Society journal primarily concerned with articles containing data on the phase behavior and the physical, thermodynamic, and transport properties of well-defined materials, including complex mixtures of known compositions. While environmental and biological samples are of interest, their compositions must be known and reproducible. As a result, adsorption on natural product materials does not generally fit within the scope of Journal of Chemical & Engineering Data.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


