Solubility Measurement and Thermodynamic Modeling of Bifendate in 13 Pure Solvents at Temperatures from 293.15 to 333.15 K
IF 2
3区 工程技术
Q3 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
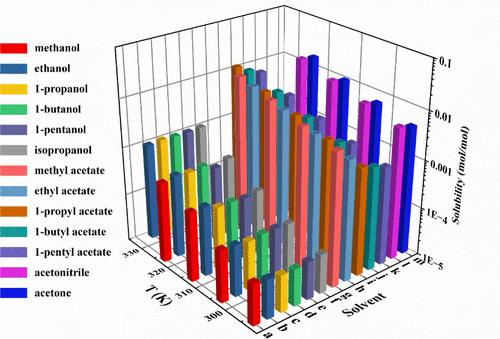
This study aimed to determine the solubility of bifendate (a drug used in the treatment of chronic hepatitis) in 13 different pure organic solvents (methanol, ethanol, 1-propanol, 1-butanol, 1-pentanol, isopropanol, methyl acetate, ethyl acetate, 1-propyl acetate, 1-butyl acetate, 1-pentyl acetate, acetonitrile, and acetone) through the gravimetric method in the range 293.15–333.15 K. The physical stability of bifendate was evaluated by comparing power X-ray diffraction patterns before and after achieving equilibrium. The melting temperature and enthalpy were measured via the differential scanning calorimetry method. These experimental data were used to regress PC-SAFT (Perturbed-Chain Statistical Associating Fluid Theory) pure component parameters of the drug. Furthermore, PC-SAFT combined with solid–liquid equilibrium theory was employed to calculate the solubility of the drug in 13 pure solvents. Combined with our previous research, this study demonstrated that PC-SAFT accurately predicted the solubility of different categories of drugs in various solvent systems. Thus, PC-SAFT was a useful tool for pharmaceutical scientists in drug research and development and could significantly save time and economic cost by reducing trial-and-error experiments.
联苯胺在 293.15 至 333.15 K 温度的 13 种纯溶剂中的溶解度测量和热力学模型建立
本研究旨在通过重量法测定联苯双酯(一种用于治疗慢性肝炎的药物)在 13 种不同纯有机溶剂(甲醇、乙醇、1-丙醇、1-丁醇、1-戊醇、异丙醇、乙酸甲酯、乙酸乙酯、乙酸 1-丙酯、乙酸 1-丁酯、乙酸 1-戊酯、乙腈和丙酮)中的溶解度,溶解度范围为 293.通过比较达到平衡前后的功率 X 射线衍射图样,评估了联苯双酯的物理稳定性。通过差示扫描量热法测量了熔化温度和焓。这些实验数据用于回归药物的 PC-SAFT(扰动链统计关联流体理论)纯组分参数。此外,还利用 PC-SAFT 结合固液平衡理论计算了该药物在 13 种纯溶剂中的溶解度。结合我们之前的研究,该研究表明 PC-SAFT 能准确预测不同类别药物在各种溶剂体系中的溶解度。因此,PC-SAFT 是制药科学家进行药物研发的有用工具,可通过减少试错实验大大节省时间和经济成本。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Chemical & Engineering Data
工程技术-工程:化工
CiteScore
5.20
自引率
19.20%
发文量
324
审稿时长
2.2 months
期刊介绍:
The Journal of Chemical & Engineering Data is a monthly journal devoted to the publication of data obtained from both experiment and computation, which are viewed as complementary. It is the only American Chemical Society journal primarily concerned with articles containing data on the phase behavior and the physical, thermodynamic, and transport properties of well-defined materials, including complex mixtures of known compositions. While environmental and biological samples are of interest, their compositions must be known and reproducible. As a result, adsorption on natural product materials does not generally fit within the scope of Journal of Chemical & Engineering Data.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


