Expanding the Design Space of Polymer–Metal Organic Framework (MOF) Gels by Understanding Polymer–MOF Interactions
IF 7.2
2区 材料科学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
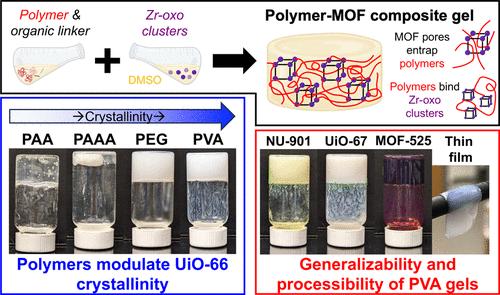
The fabrication of polymer-MOF composite gels holds great potential to provide emergent properties for drug delivery, environmental remediation, and catalysis. To leverage the full potential of these composites, we investigated how the presence and chemistry of polymers impact MOF formation within the composites and, in turn, how MOFs impact polymer gelation. We show that polymers with a high density of strongly metal-binding carboxylic acids inhibit MOF formation; however, reducing the density of carboxylic acids or substituting them with weaker metal-binding hydroxyl groups permits both MOF formation and gelation within composites. Preparing composites with poly(ethylene glycol) (PEG), which does not bind MOF zirconium (Zr)-oxo clusters, and observing gelation suggests that MOFs can entrap polymer chains to create cross-links in addition to cross-linking them through polymer-Zr-oxo interactions. Both simulations and experiments show composite hydrogels formed with poly(vinyl alcohol) (PVA) to be more stable than those made with PEG, which can reptate through MOF pores upon heating. We demonstrate the generalizability of this composite formation process across different Zr-based MOFs (UiO-66, NU-901, UiO-67, and MOF-525) and by spin-coating gels into conformable films. PVA-UiO-66 composite hydrogels demonstrated high sorption and sustained release of methylene blue relative to the polymer alone (3× loading, 28× slower release), and PVA-MOF-525 composite hydrogels capably sorb the therapeutic peptide Angiotensin 1–7. By understanding the influence of polymer-MOF interactions on the structure and properties of composite gels, this work informs and expands the design space of this emerging class of materials.
通过了解聚合物与金属有机框架 (MOF) 的相互作用拓展聚合物-金属有机框架凝胶的设计空间
聚合物-MOF 复合凝胶的制造具有巨大潜力,可为药物输送、环境修复和催化提供新兴特性。为了充分发挥这些复合材料的潜力,我们研究了聚合物的存在和化学性质如何影响复合材料中 MOF 的形成,以及 MOF 反过来又如何影响聚合物凝胶化。我们的研究表明,具有高密度强金属结合羧酸的聚合物会抑制 MOF 的形成;然而,降低羧酸的密度或用金属结合力较弱的羟基取代羧酸,就能在复合材料中形成 MOF 并使其凝胶化。用聚乙二醇(PEG)制备复合材料(PEG 不会结合 MOF 锆(Zr)-氧代簇)并观察凝胶化现象表明,除了通过聚合物-Zr-氧代相互作用交联外,MOF 还可以夹带聚合物链以产生交联。模拟和实验都表明,用聚(乙烯醇)(PVA)形成的复合水凝胶比用聚乙二醇(PEG)形成的复合水凝胶更稳定,因为后者在加热时会重新通过 MOF 孔隙。我们证明了这种复合材料形成过程在不同的 Zr 基 MOF(UiO-66、NU-901、UiO-67 和 MOF-525)上的通用性,并通过旋涂凝胶使其成为适形薄膜。与单独使用聚合物相比,PVA-UiO-66 复合水凝胶对亚甲蓝具有较高的吸附性和持续释放性(负载量增加 3 倍,释放速度减慢 28 倍),PVA-MOF-525 复合水凝胶可吸附治疗肽血管紧张素 1-7。通过了解聚合物-MOF 相互作用对复合凝胶结构和性能的影响,这项研究成果为这一新兴材料类别的设计提供了信息并拓展了设计空间。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemistry of Materials
工程技术-材料科学:综合
CiteScore
14.10
自引率
5.80%
发文量
929
审稿时长
1.5 months
期刊介绍:
The journal Chemistry of Materials focuses on publishing original research at the intersection of materials science and chemistry. The studies published in the journal involve chemistry as a prominent component and explore topics such as the design, synthesis, characterization, processing, understanding, and application of functional or potentially functional materials. The journal covers various areas of interest, including inorganic and organic solid-state chemistry, nanomaterials, biomaterials, thin films and polymers, and composite/hybrid materials. The journal particularly seeks papers that highlight the creation or development of innovative materials with novel optical, electrical, magnetic, catalytic, or mechanical properties. It is essential that manuscripts on these topics have a primary focus on the chemistry of materials and represent a significant advancement compared to prior research. Before external reviews are sought, submitted manuscripts undergo a review process by a minimum of two editors to ensure their appropriateness for the journal and the presence of sufficient evidence of a significant advance that will be of broad interest to the materials chemistry community.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


