In Vitro Osteo-Immunological Responses of Bioactive Calcium Phosphate-Containing Urethane Dimethacrylate-Based Composites: A Potential Alternative to Poly(methyl methacrylate) Bone Cement
IF 5.7
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
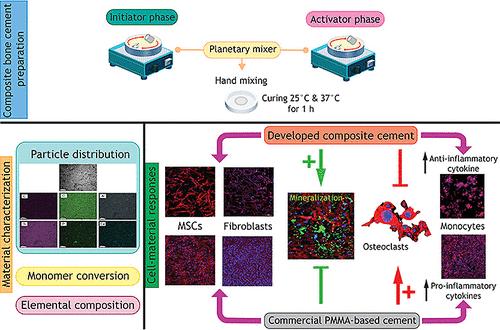
This investigation developed new composite bone cements using urethane dimethacrylate (UDMA), poly(propylene glycol) dimethacrylate (PPGDMA), and hydroxyethyl methacrylate (HEMA), with micrometer-sized aluminosilicate glass filler. Monocalcium phosphate monohydrate (MCPM) and hydroxyapatite (HA) particles were added to enhance biological performance, particularly osteo-immunomodulation. Free radical polymerization was triggered by mixing two pastes containing either benzoyl peroxide (BPO, an initiator) or N-tolyglycine glycidyl methacrylate (NTGGMA, an activator). Increasing butylated hydroxytoluene (BHT, an inhibitor) enabled a suitable delay after mixing at 25 °C for placement. At 37 °C, the delay time was reduced and the final conversion was enhanced. Findings also demonstrated the biocompatibility of the developed bone cement toward osteo-immunological cell lineages, including mesenchymal stem cells (MSCs), fibroblasts, osteoclast precursor RAW 246.7 cells, and peripheral blood mononuclear cells (PBMCs). Notably, the cement with both MCPM and HA combined facilitated sufficient MSC growth, enabling subsequent mineralization while concurrently suppressing the proliferation of fibroblasts, osteoclast progenitors, and PBMCs. Furthermore, composite cement exhibited the capacity to differentially regulate osteoblast differentiation, cell-(in)dependent mineralization, osteoclastogenesis, and PBMC-mediated inflammatory responses at both cellular and molecular levels in vitro. These observations suggested their potential use for bone repair, especially in cases of inflammation-associated bone defects.
含生物活性磷酸钙的聚氨酯二甲基丙烯酸酯基复合材料的体外骨免疫反应:聚甲基丙烯酸甲酯骨水泥的潜在替代品
这项研究使用聚氨酯二甲基丙烯酸酯(UDMA)、聚丙二醇二甲基丙烯酸酯(PPGDMA)和甲基丙烯酸羟乙酯(HEMA)以及微米级硅酸铝玻璃填料开发了新型复合骨水泥。此外,还添加了一水磷酸一钙(MCPM)和羟基磷灰石(HA)颗粒,以提高生物性能,尤其是骨免疫调节性能。将两种含有过氧化苯甲酰(BPO,一种引发剂)或 N-聚甘氨酸缩水甘油醚甲基丙烯酸酯(NTGGMA,一种活化剂)的浆料混合,可引发自由基聚合。增加丁基羟基甲苯(BHT,一种抑制剂)可在 25 °C下混合后适当延迟放置时间。37 °C时,延迟时间缩短,最终转化率提高。研究结果还证明了开发的骨水泥对骨免疫细胞系的生物相容性,包括间充质干细胞(MSC)、成纤维细胞、破骨细胞前体 RAW 246.7 细胞和外周血单核细胞(PBMC)。值得注意的是,含有 MCPM 和 HA 的复合水泥能促进间叶干细胞的充分生长,使其随后矿化,同时抑制成纤维细胞、破骨细胞前体细胞和外周血单核细胞的增殖。此外,复合骨水泥还能在体外的细胞和分子水平上对成骨细胞分化、细胞(内)依赖性矿化、破骨细胞生成和 PBMC 介导的炎症反应进行不同程度的调节。这些观察结果表明,它们有可能用于骨修复,尤其是炎症相关的骨缺损。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Materials Au
材料科学-
CiteScore
5.00
自引率
0.00%
发文量
0
期刊介绍:
ACS Materials Au is an open access journal publishing letters articles reviews and perspectives describing high-quality research at the forefront of fundamental and applied research and at the interface between materials and other disciplines such as chemistry engineering and biology. Papers that showcase multidisciplinary and innovative materials research addressing global challenges are especially welcome. Areas of interest include but are not limited to:Design synthesis characterization and evaluation of forefront and emerging materialsUnderstanding structure property performance relationships and their underlying mechanismsDevelopment of materials for energy environmental biomedical electronic and catalytic applications
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


