Liquid−Liquid Equilibrium Data for 1-Butanol–Methylcyclohexane and Acetonitrile–Benzene Mixtures Using Multiple Deep Eutectic Solvents Based on Choline Chloride
IF 2
3区 工程技术
Q3 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
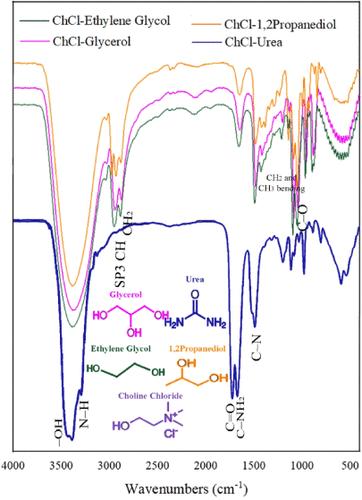
Choline chloride (ChCl)-based deep eutectic solvents (DESs) have been utilized as a green extracting agent for the efficient extraction of 1-butanol from methylcyclohexane + butanol combinations and acetonitrile from benzene + acetonitrile mixture at atmospheric pressure and 298.15 K. The prepared DESs were ChCl + urea (DES1; 1:2 molar ratio), ChCl + 1,2-propanediol (DES2; 1:3 molar ratio), ChCl + ethylene glycol (DES3; 1:2 molar ratio), and ChCl + glycerol (DES4; 1:2 molar ratio). The Hand and Othmer–Tobias equations were used to evaluate the tie-line compositions’ reliability. The extrication abilities of the examined DESs to separate methylcyclohexane +1-butanol and benzene + acetonitrile mixtures were compared to the NRTL model. The binary interaction parameters were established when the NRTL model corroborated the measured equilibrium data. The Gibbs topological analysis was utilized to validate consistency of the model parameters. The extraction equilibrium data showed that the selectivity parameter was higher than unity (S > 1) in all the evaluated systems. The distribution coefficient factor in systems containing ChCl–urea was higher than unity, indicating the need for smaller amounts of solvents to separate 1-butanol from methylcyclohexane, and the number of extraction steps was reduced. The distribution coefficient factor was less than unity in all systems, and more DESs were needed for optimal separation of acetonitrile from benzene.
使用基于氯化胆碱的多种深共晶溶剂的 1-丁醇-甲基环己烷和乙腈-苯混合物的液液平衡数据
基于氯化胆碱(ChCl)的深共晶溶剂(DES)被用作一种绿色萃取剂,在常压和298.15 K条件下从甲基环己烷+丁醇组合物中高效萃取1-丁醇,从苯+乙腈混合物中高效萃取乙腈。制备的 DES 分别为氯化 ChCl + 尿素(DES1;摩尔比 1:2)、氯化 ChCl + 1,2-丙二醇(DES2;摩尔比 1:3)、氯化 ChCl + 乙二醇(DES3;摩尔比 1:2)和氯化 ChCl + 甘油(DES4;摩尔比 1:2)。采用汉德方程和 Othmer-Tobias 方程来评估扎线成分的可靠性。将所研究的 DES 分离甲基环己烷+1-丁醇和苯+乙腈混合物的能力与 NRTL 模型进行了比较。当 NRTL 模型与测得的平衡数据相吻合时,二元相互作用参数就确定了。吉布斯拓扑分析被用来验证模型参数的一致性。萃取平衡数据显示,在所有被评估的体系中,选择性参数都高于统一值(S >1)。在含有 ChCl-脲的体系中,分配系数系数高于统一值,这表明从甲基环己烷中分离 1-丁醇需要较少的溶剂,萃取步骤的数量也减少了。所有系统中的分配系数均小于统一值,因此需要更多的 DES 才能从苯中分离出乙腈。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Chemical & Engineering Data
工程技术-工程:化工
CiteScore
5.20
自引率
19.20%
发文量
324
审稿时长
2.2 months
期刊介绍:
The Journal of Chemical & Engineering Data is a monthly journal devoted to the publication of data obtained from both experiment and computation, which are viewed as complementary. It is the only American Chemical Society journal primarily concerned with articles containing data on the phase behavior and the physical, thermodynamic, and transport properties of well-defined materials, including complex mixtures of known compositions. While environmental and biological samples are of interest, their compositions must be known and reproducible. As a result, adsorption on natural product materials does not generally fit within the scope of Journal of Chemical & Engineering Data.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


