A bispecific nanosystem activates endogenous natural killer cells in the bone marrow for haematologic malignancies therapy
IF 38.1
1区 材料科学
Q1 MATERIALS SCIENCE, MULTIDISCIPLINARY
引用次数: 0
Abstract
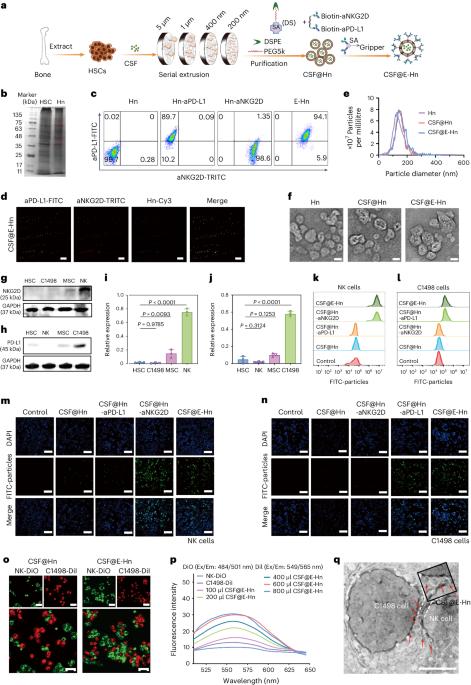
Haematologic malignancies commonly arise from the bone marrow lesion, yet there are currently no effective targeted therapies against tumour cells in this location. Here we constructed a bone-marrow-targeting nanosystem, CSF@E-Hn, which is based on haematopoietic-stem-cell-derived nanovesicles adorned with gripper ligands (aPD-L1 and aNKG2D) and encapsulated with colony-stimulating factor (CSF) for the treatment of haematologic malignancies. CSF@E-Hn targets the bone marrow and, thanks to the gripper ligands, pulls together tumour cells and natural killer cells, activating the latter for specific tumour cell targeting and elimination. The therapeutic efficacy was validated in mice bearing acute myeloid leukaemia and multiple myeloma. The comprehensive assessment of the post-treatment bone marrow microenvironment revealed that the integration of CSF into a bone-marrow-targeted nanosystem promoted haematopoietic stem cell differentiation, boosted memory T cell generation and maintained bone homoeostasis, with long-term prevention of relapse. Our nanosystem represents a promising strategy for the treatment of haematologic malignancies. Blood cancers generally derive from the bone marrow but there are no current targeted treatments. Here the authors present a bone-marrow-targeting nanoparticle that can bind and pull together tumour and natural killer cells for selective elimination of cancer cells, offering a strategy for the treatment of haematologic cancers.
一种双特异性纳米系统可激活骨髓中的内源性自然杀伤细胞,用于治疗血液恶性肿瘤。
血液恶性肿瘤通常来自骨髓病变,但目前还没有针对该部位肿瘤细胞的有效靶向疗法。在这里,我们构建了一种骨髓靶向纳米系统 CSF@E-Hn,它基于造血干细胞衍生的纳米颗粒,颗粒上装饰有抓取配体(aPD-L1 和 aNKG2D),并包裹有集落刺激因子(CSF),用于治疗血液恶性肿瘤。CSF@E-Hn以骨髓为靶点,通过抓取配体将肿瘤细胞和自然杀伤细胞聚集在一起,激活后者进行特异性肿瘤细胞靶向清除。在罹患急性髓性白血病和多发性骨髓瘤的小鼠身上验证了其疗效。对治疗后骨髓微环境的综合评估显示,将 CSF 纳入骨髓靶向纳米系统可促进造血干细胞分化,促进记忆 T 细胞的生成,维持骨平衡,长期预防复发。我们的纳米系统是治疗血液恶性肿瘤的一种前景广阔的策略。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature nanotechnology
工程技术-材料科学:综合
CiteScore
59.70
自引率
0.80%
发文量
196
审稿时长
4-8 weeks
期刊介绍:
Nature Nanotechnology is a prestigious journal that publishes high-quality papers in various areas of nanoscience and nanotechnology. The journal focuses on the design, characterization, and production of structures, devices, and systems that manipulate and control materials at atomic, molecular, and macromolecular scales. It encompasses both bottom-up and top-down approaches, as well as their combinations.
Furthermore, Nature Nanotechnology fosters the exchange of ideas among researchers from diverse disciplines such as chemistry, physics, material science, biomedical research, engineering, and more. It promotes collaboration at the forefront of this multidisciplinary field. The journal covers a wide range of topics, from fundamental research in physics, chemistry, and biology, including computational work and simulations, to the development of innovative devices and technologies for various industrial sectors such as information technology, medicine, manufacturing, high-performance materials, energy, and environmental technologies. It includes coverage of organic, inorganic, and hybrid materials.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


