Regulation of Absorption and Emission in a Protein/Fluorophore Complex.
IF 3.5
2区 生物学
Q2 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Human cellular retinol binding protein II (hCRBPII) was used as a protein engineering platform to rationally regulate absorptive and emissive properties of a covalently bound fluorogenic dye. We demonstrate the binding of a thio-dapoxyl analog via formation of a protonated imine between an active site lysine residue and the chromophore's aldehyde. Rational manipulation of the electrostatics of the binding pocket results in a 204 nm shift in absorption and a 131 nm shift in emission. The protein is readily expressed in mammalian systems and binds with exogenously delivered fluorophore as demonstrated by live-cell imaging experiments.
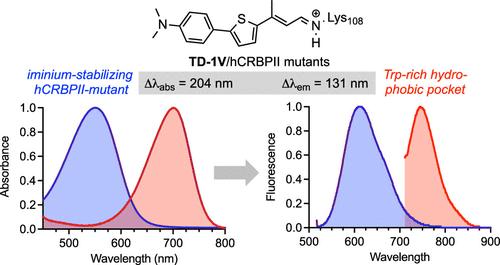
调节蛋白质/荧光团复合物的吸收和发射。
人类细胞视黄醇结合蛋白 II(hCRBPII)被用作一个蛋白质工程平台,用于合理调节共价结合荧光染料的吸收和发射特性。我们通过在活性位点赖氨酸残基和发色团的醛之间形成质子化的亚胺,证明了硫代二乙酰类似物的结合。对结合袋的静电进行合理操作后,吸收波长发生了 204 nm 的移动,发射波长发生了 131 nm 的移动。活细胞成像实验证明,这种蛋白质很容易在哺乳动物系统中表达,并能与外源性荧光团结合。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Chemical Biology
生物-生化与分子生物学
CiteScore
7.50
自引率
5.00%
发文量
353
审稿时长
3.3 months
期刊介绍:
ACS Chemical Biology provides an international forum for the rapid communication of research that broadly embraces the interface between chemistry and biology.
The journal also serves as a forum to facilitate the communication between biologists and chemists that will translate into new research opportunities and discoveries. Results will be published in which molecular reasoning has been used to probe questions through in vitro investigations, cell biological methods, or organismic studies.
We welcome mechanistic studies on proteins, nucleic acids, sugars, lipids, and nonbiological polymers. The journal serves a large scientific community, exploring cellular function from both chemical and biological perspectives. It is understood that submitted work is based upon original results and has not been published previously.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


