Structural Engineering of l-Aspartic Amphiphilic Polyesters for Enzyme-Responsive Drug Delivery and Bioimaging in Cancer Cells
IF 4.7
Q1 POLYMER SCIENCE
引用次数: 0
Abstract
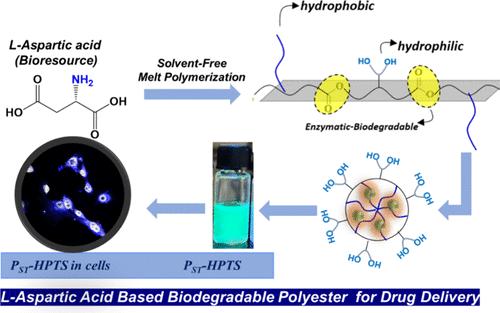
Design and development of amphiphilic polyesters based on bioresources are very important to cater to the ever-growing need for biodegradable polymers in biomedical applications. Here, we report structural engineering of enzyme-responsive amphiphilic polyesters based on l-amino acid bioresources and study their drug delivery aspects in the cancer cell line. For this purpose, an l-aspartic acid-based polyester platform is chosen, and two noncovalent forces such as hydrogen bonding and side-chain hydrophobic interactions are introduced to study their effect on the aqueous self-assembly of nanoparticles. The synthetic strategy involves the development of l-aspartic acid-based dimethyl ester monomers with acetal and stearate side chains and subjecting them to solvent-free melt polycondensation reactions to produce side-chain-functionalized polyesters in the entire composition range. Postpolymerization acid catalyst deprotection of acetal yielded hydroxyl-functionalized polyesters. Amphiphilicity of the polymer is carefully fine-tuned by varying the composition of the stearate and hydroxyl units in the polymer chains to produce self-assembly in water. Various drugs such as camptothecin (CPT), curcumin (CUR), and doxorubicin (DOX) and biomarkers like 8-hydroxypyrene-1,3,6-trisulfonic acid trisodium salt (HPTS), rose bengal (RB), and Nile red (NR) are successfully encapsulated in the polymer nanoparticles. Cytotoxicity of biodegradable polymer nanoparticles is tested in normal and breast cancer cell lines. The polymer nanoparticles are found to be highly biocompatible and delivered the anticancer drugs in the intracellular compartments of the cells.
用于癌细胞酶反应性给药和生物成像的 l-天冬氨酸两性聚酯的结构工程学研究
基于生物资源的两亲性聚酯的设计和开发对于满足生物医学应用中对可生物降解聚合物日益增长的需求非常重要。在此,我们报告了基于 l- 氨基酸生物资源的酶响应性两亲性聚酯的结构工程,并研究了它们在癌细胞系中的药物输送方面。为此,我们选择了一种基于 l-天冬氨酸的聚酯平台,并引入了两种非共价作用力,如氢键和侧链疏水相互作用,以研究它们对纳米粒子水性自组装的影响。合成策略包括开发具有缩醛和硬脂酸侧链的 l-天冬氨酸基二甲酯单体,并将其进行无溶剂熔融缩聚反应,以生产整个组成范围内的侧链官能化聚酯。聚合后酸催化剂对缩醛进行脱保护,得到羟基官能化聚酯。通过改变聚合物链中硬脂酸单元和羟基单元的组成,可对聚合物的亲水性进行细致的微调,从而在水中产生自组装。喜树碱 (CPT)、姜黄素 (CUR) 和多柔比星 (DOX) 等多种药物以及 8-羟基芘-1,3,6-三磺酸三钠盐 (HPTS)、玫瑰红 (RB) 和尼罗红 (NR) 等生物标记物被成功封装在聚合物纳米颗粒中。在正常细胞系和乳腺癌细胞系中测试了生物可降解聚合物纳米粒子的细胞毒性。结果发现,这种聚合物纳米粒子具有很高的生物相容性,能将抗癌药物输送到细胞内。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


