Thermodynamics and Kinetics of Atmospherically Relevant Acetalization Reactions
IF 2.9
3区 化学
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
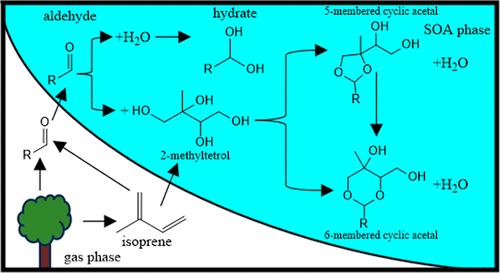
Accretion reactions have been suggested as an important mechanism in the formation of low volatility secondary organic aerosol (SOA). Acetals are potential accretion products formed through the acid-catalyzed reactions of aldehydes and alcohols (both of which are ubiquitous in the atmosphere) via nucleophilic addition of alcohols to the aldehyde C═O bond. The thermodynamics and kinetics of the acetalization reaction mechanism for model aldehydes and polyols were examined with computational electronic structure methods and experimental bulk kinetics experiments using nuclear magnetic resonance (NMR) spectroscopy. The formation of all possible nucleophilic addition reaction products (hydrates, hemiacetals, acyclic acetals, and cyclic acetals) was found to be thermodynamically feasible under aqueous acidic conditions, a result at odds with an earlier computational study. Except for the formation of the thermodynamically most favored cyclic acetal products, the reactions occurred on a time scale shorter than the NMR experiment (lifetimes <5 min). Polyols that could form both a 5- or 6-membered ring cyclic acetal product formed the 5-membered ring faster but exhibited interconversion to the 6-membered ring on the multihour lifetime scale. Reactions involving structurally different aldehydes and polyols were also examined, but both the thermodynamics and kinetics results depended only weakly on the carbon backbone of reactant aldehydes and polyols. Even the relatively slow cyclic acetal-forming reactions were found to have lifetimes <1 day for pH 2.2 SOA acidities which are well within atmospherically relevant time scales. These thermodynamic and kinetic results indicate that acetalization reactions are a plausible accretion mechanism in SOA.
与大气有关的乙缩醛反应的热力学和动力学
吸积反应被认为是低挥发性二次有机气溶胶(SOA)形成的一个重要机制。醛和醇(这两种物质在大气中无处不在)在酸催化下发生反应,通过醇与醛的 C═O 键发生亲核加成反应而形成乙缩醛,乙缩醛是潜在的增殖产物。通过计算电子结构方法和使用核磁共振 (NMR) 光谱法进行的体动力学实验,研究了模型醛和多元醇缩醛化反应机制的热力学和动力学。研究发现,在酸性水溶液条件下,所有可能的亲核加成反应产物(水合物、半乙缩醛、无环乙缩醛和环乙缩醛)的形成在热力学上都是可行的,这一结果与早先的计算研究不一致。除了形成热力学上最有利的环缩醛产物外,反应发生的时间尺度比核磁共振实验要短(寿命为 5 分钟)。同时可以形成 5 元环或 6 元环环状缩醛产物的多元醇形成 5 元环的速度更快,但在多小时的寿命范围内表现出向 6 元环的相互转化。还研究了涉及结构不同的醛和多元醇的反应,但热力学和动力学结果对反应物醛和多元醇的碳骨架的依赖性都很弱。在 pH 值为 2.2 的 SOA 酸性条件下,即使是相对缓慢的环状缩醛生成反应,其寿命也只有 1 天,完全符合大气相关的时间尺度。这些热力学和动力学结果表明,缩醛化反应是 SOA 中一种可信的增殖机制。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Earth and Space Chemistry
Earth and Planetary Sciences-Geochemistry and Petrology
CiteScore
5.30
自引率
11.80%
发文量
249
期刊介绍:
The scope of ACS Earth and Space Chemistry includes the application of analytical, experimental and theoretical chemistry to investigate research questions relevant to the Earth and Space. The journal encompasses the highly interdisciplinary nature of research in this area, while emphasizing chemistry and chemical research tools as the unifying theme. The journal publishes broadly in the domains of high- and low-temperature geochemistry, atmospheric chemistry, marine chemistry, planetary chemistry, astrochemistry, and analytical geochemistry. ACS Earth and Space Chemistry publishes Articles, Letters, Reviews, and Features to provide flexible formats to readily communicate all aspects of research in these fields.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


