A methodological pipeline for the preclinical evaluation of novel topical agents for the treatment of CRS
IF 7.2
2区 医学
Q1 OTORHINOLARYNGOLOGY
引用次数: 0
Abstract
Key points
- Novel topical therapeutics require extensive pre-clinical testing to assess efficacy and safety.
- Antibiofilm or immunosuppressant agents can utilize ex vivo models to measure ciliotoxicity.
- Agents that are found to be effective and non-toxic ex vivo warrant further investigation in vivo.
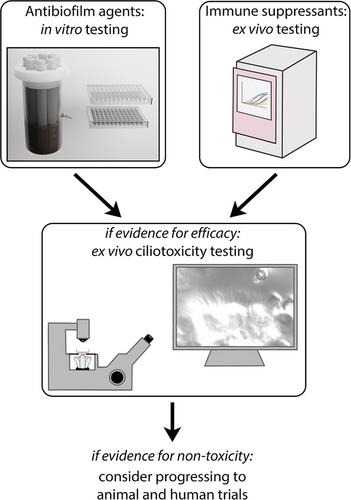
用于治疗 CRS 的新型外用制剂临床前评估的方法流水线。
要点:新型局部治疗药物需要进行广泛的临床前测试,以评估疗效和安全性。抗生物膜或免疫抑制剂可利用体外模型来衡量纤毛毒性。在体外发现有效且无毒的制剂应在体内进行进一步研究。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

International Forum of Allergy & Rhinology
OTORHINOLARYNGOLOGY-
CiteScore
11.70
自引率
10.90%
发文量
185
审稿时长
6-12 weeks
期刊介绍:
International Forum of Allergy & Rhinologyis a peer-reviewed scientific journal, and the Official Journal of the American Rhinologic Society and the American Academy of Otolaryngic Allergy.
International Forum of Allergy Rhinology provides a forum for clinical researchers, basic scientists, clinicians, and others to publish original research and explore controversies in the medical and surgical treatment of patients with otolaryngic allergy, rhinologic, and skull base conditions. The application of current research to the management of otolaryngic allergy, rhinologic, and skull base diseases and the need for further investigation will be highlighted.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


