Revealing new pathways for the reaction of Criegee intermediate CH2OO with SO2
IF 5.9
2区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
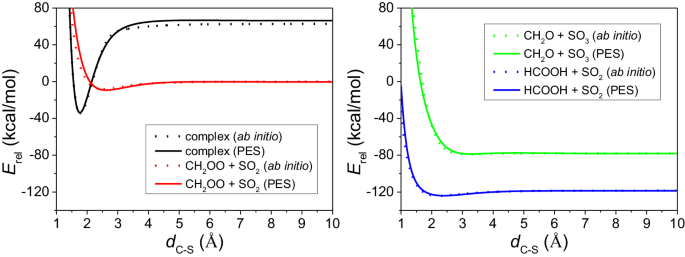
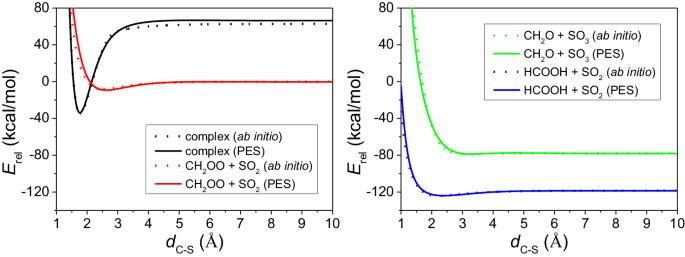
Criegee intermediates play an important role in the tropospheric oxidation models through their reactions with atmospheric trace chemicals. We develop a global full-dimensional potential energy surface for the CH2OO + SO2 system and reveal how the reaction happens step by step by quasi-classical trajectory simulations. A new pathway forming the main products (CH2O + SO3) and a new product channel (CO2 + H2 + SO2) are predicted in our simulations. The new pathway appears at collision energies greater than 10 kcal/mol whose behavior demonstrates a typical barrier-controlled reaction. This threshold is also consistent with the ab initio transition state barrier height. For the minor products, a loose complex OCH2O ∙ ∙ ∙ SO2 is formed first, and then in most cases it soon turns into HCOOH + SO2, in a few cases it decomposes into CO2 + H2 + SO2 which is a new product channel, and rarely it remains as ∙OCH2O ∙ + SO2. Criegee intermediates such as CH2OO play an important role in tropospheric oxidation models through their reactions with atmospheric trace chemicals. Here, the authors develop a global full-dimensional potential energy surface for the CH2OO + SO2 system, reveal how the reaction happens step by step using quasi-classical trajectory simulations, to show a new direct stripping pathway forming the main products CH2O and SO3 and a new product channel.
揭示克里基中间体 CH2OO 与二氧化硫反应的新途径。
克里基中间体通过与大气中的痕量化学物质发生反应,在对流层氧化模型中发挥着重要作用。我们为 CH2OO + SO2 系统开发了一个全局全维势能面,并通过准经典轨迹模拟揭示了反应是如何一步步发生的。我们在模拟中预测了形成主要产物(CH2O + SO3)的新途径和新产物通道(CO2 + H2 + SO2)。新通道出现在碰撞能量大于 10 kcal/mol 的情况下,其行为表现为典型的势垒控制反应。这一阈值也与 ab initio 过渡态势垒高度相一致。对于次要产物,首先会形成一个松散的复合物 OCH2O ∙∙ ∙ SO2,然后在大多数情况下很快会变成 HCOOH + SO2,在少数情况下会分解成 CO2 + H2 + SO2,这是新的产物途径,而在极少数情况下会保持为 ∙OCH2O ∙ + SO2。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Communications Chemistry
Chemistry-General Chemistry
CiteScore
7.70
自引率
1.70%
发文量
146
审稿时长
13 weeks
期刊介绍:
Communications Chemistry is an open access journal from Nature Research publishing high-quality research, reviews and commentary in all areas of the chemical sciences. Research papers published by the journal represent significant advances bringing new chemical insight to a specialized area of research. We also aim to provide a community forum for issues of importance to all chemists, regardless of sub-discipline.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


