Enantioselective Michael addition of malonates to α,β-unsaturated ketones catalyzed by rare-earth metal amides RE[N(SiMe3)2]3 with phenoxy-functionalized amino alcohol proligands
IF 1.5
4区 化学
Q3 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
A combination of rare-earth metal amides RE[N(SiMe3)2]3 and chiral phenoxy-functionalized amino alcohol proligands was developed to realize the enantioselective Michael addition of malonates with unsaturated ketones. The reactions catalyzed by samarium amide Sm[N(SiMe3)2]3 were performed best together with the chiral proligand H3L1 (H3L1 = 2,4-di-tert-butyl-6-((((1S,2R)-2-hydroxy-1,2-diphenylethyl) amino)methyl)phenol) in DCE with good group tolerance, mostly in high to excellent yields (85–98 %) and good to excellent ee values (50− >99 %). The possible mechanism was also proposed.
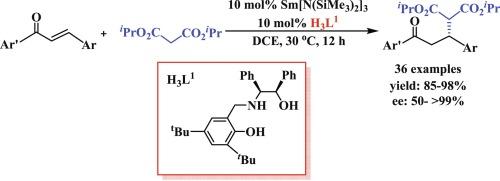
稀土金属酰胺 RE[N(SiMe3)2]3 与苯氧基官能化氨基醇原配体催化的丙二酸盐与α、β-不饱和酮的对映选择性迈克尔加成反应
研究人员开发了一种稀土金属酰胺 RE[N(SiMe3)2]3 与手性苯氧基官能化氨基醇原配体的组合,以实现丙二酸盐与不饱和酮的对映选择性迈克尔加成反应。钐酰胺 Sm[N(SiMe3)2]3 与手性原配体 H3L1(H3L1 = 2,4-二叔丁基-6-((((1S,2R)-2-羟基-1,2-二苯基)氨基甲基苯酚)在二氯乙烷中催化的反应具有良好的基团耐受性,大部分反应的产率从高到优(85-98 %),ee值从好到优(50-99 %)。还提出了可能的机理。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Tetrahedron Letters
化学-有机化学
CiteScore
3.50
自引率
5.60%
发文量
521
审稿时长
28 days
期刊介绍:
Tetrahedron Letters provides maximum dissemination of outstanding developments in organic chemistry. The journal is published weekly and covers developments in techniques, structures, methods and conclusions in experimental and theoretical organic chemistry. Rapid publication of timely and significant research results enables researchers from all over the world to transmit quickly their new contributions to large, international audiences.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


