Electrochemical oxidations through hypervalent iodine redox catalysis
引用次数: 0
Abstract
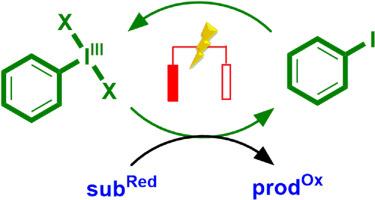
In recent years, a growing number of oxidations that involve electrochemical regeneration of catalytic hypervalent iodine species has been developed. These transformations benefit from the combination of rich reactivity offered by hypervalent iodine compounds and sustainability advantage provided by electrocatalysis, which eliminates the need for any stoichiometric redox reagents. This review provides a systematic overview of this emerging field, covering the most abundant two-electron electrocatalysis with iodoarenes via I(I)/I(III) redox cycle, the isolated existing example of I(V)/I(VII) cycle, and a newly developed approach exploiting iodoarenes as I(I)/I(II) single-electron transfer mediators.
通过超价碘氧化还原催化实现电化学氧化
近年来,越来越多涉及催化超价碘物种电化学再生的氧化技术被开发出来。超价碘化合物具有丰富的反应活性,而电催化技术则无需使用任何化学计量氧化还原试剂,因而具有可持续发展的优势。本综述对这一新兴领域进行了系统概述,涵盖了通过 I(I)/I(III) 氧化还原循环利用碘代烯烃进行的最丰富的双电子电催化、I(V)/I(VII) 循环的孤立实例,以及利用碘代烯烃作为 I(I)/I(II) 单电子转移介质的新开发方法。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


