Electrochemical oxidative thioetherification of aldehyde hydrazones with thiophenols†
IF 2.9
3区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
An electrochemically promoted oxidative dehydrogenation cross-coupling reaction between aldehyde hydrazones and thiophenols is demonstrated for the first time, which resulted in a variety of (Z)-thioetherified products in moderate to excellent yields. This strategy can be carried out under an air atmosphere, featuring scalability and excellent stereoselectivity. In addition, the transformation efficiently produces readily recyclable disulfide as a by-product with high yields, which significantly reduces the environmental pollution caused by thioetherification.
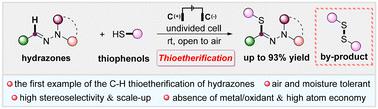
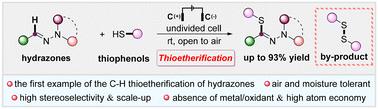
醛肼与噻吩酚的电化学氧化硫醚化反应。
该研究首次展示了醛肼和噻吩酚之间的电化学促进氧化脱氢交叉偶联反应,该反应以中等至优异的产率产生了多种 (Z) -硫醚化产物。该方法可在空气气氛下进行,具有可扩展性和出色的立体选择性。此外,这种转化方法还能以高产率有效地产生易于回收的二硫化物作为副产品,从而大大减少了硫醚化反应对环境造成的污染。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Organic & Biomolecular Chemistry
化学-有机化学
CiteScore
5.50
自引率
9.40%
发文量
1056
审稿时长
1.3 months
期刊介绍:
The international home of synthetic, physical and biomolecular organic chemistry.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


