Reduced TiO2 nanotube array electrode based supercapacitor with kilohertz frequency response
Abstract
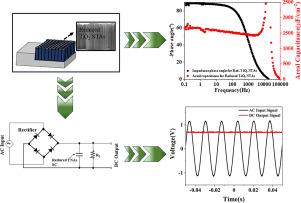
Enhancing the frequency of traditional supercapacitors to hundreds or thousands of Hz enables them to replace bulky aluminum electrolytic capacitors for line filtering and function as storage device for the harnessed ambient noise energy for powering the distributed sensor networks and IoT. This work reports a kHz frequency-capable pseudocapacitor comprising electrodes with anatase nanotube arrays (NTA). NTA are grown in-situ via anodization of a titanium foil, providing excellent electrical contact with the underlying unconverted titanium foil. The use of an organic electrolyte (glycerol and ethylene glycol solvent) allows greater control over NTA growth and enables fine-tuning of morphology. Electrochemical reduction of the NTA significantly lowers electrode resistance, thereby enhancing oxygen vacancies and leading to a two-order-of-magnitude rise in charge carrier density (from 2.20 × 1019 cm−3 to 1.03 × 1021 cm−3), as determined by Mott-Schottky analysis. The electrode exhibits a high areal capacitance of 1517 F cm−2 and a phase angle of at 120 Hz. This performance compares favorably with most carbon-based kHz supercapacitor electrodes. The upper-frequency limit of operation for the pseudocapacitor, as measured by the self-resonance frequency, is a high value of 80 kHz.

| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


