Anomalous friction of confined water in carbon nanotubes
Abstract
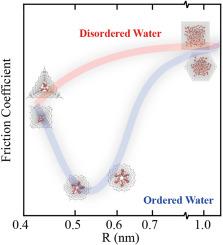
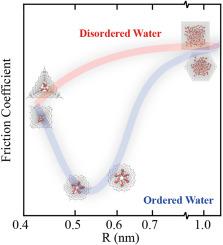
The friction of water within a carbon nanotube (CNT) is influenced by the interplay between energy barriers and water structure. In this work, we employ a series of regular polygonal CNTs, whose energy barriers remain constant with size, to examine the influence of water structure on solid-water friction using the molecular dynamics (MD) method. Polygonal CNTs with radii under 0.45 nm show friction coefficients an order of magnitude higher than their circular counterparts. While water exhibits an ordered phase within 0.5–0.6 nm-radius polygonal CNTs, resulting in a significant 80 % reduction in the friction coefficients compared to bulk like water. The force distribution analysis confirms the constancy of energy barriers. Further analysis of water density, hydrogen bond number distribution, average structure factor, and density correlation time demonstrates that the density correlation time predominantly impacts solid-liquid friction. The observed reduction in friction is primarily due to the collective movement of water molecules in an ordered arrangement. These findings illuminate nanoscale drag reduction mechanisms, offering insights for micro-nano flow system design.

| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


