Environmentally Friendly Recovery of Li2CO3 from Spent Lithium-Ion Batteries by Oxidation and Selective Leaching Process
IF 7.4
Q1 ENGINEERING, ENVIRONMENTAL
引用次数: 0
Abstract
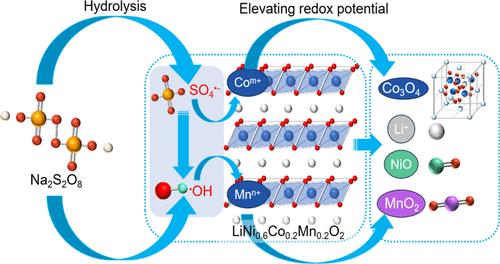
The extraction of valuable metals from spent Ni–Co–Mn oxide (NCM) cathodes typically encounters the use of strong acids or alkalis, often leading to secondary pollution. Herein, an environmentally friendly recovery route for the selective extraction of lithium (Li) by using sodium persulfate (Na2S2O8) as the sole leaching agent was proposed. Under the optimized conditions, the leaching efficiency of Li achieved 98.02%, and the selective leaching efficiency of Li was 94.80%. Moreover, the lithium carbonate (Li2CO3) product was recovered from the Li-rich filtrate with a high purity of 99.5%. The mechanism of Li selective leaching was revealed by means of wet chemistry, kinetics, thermodynamics, and solid-phase analysis. During selective leaching, free radicals SO4•– and •OH, hydron ion (H+), and sodium ion (Na+) were generated by Na2S2O8. These free radicals can increase the redox potential of the leaching system. Under these conditions, Co and Mn elements were both maintained in a high valence state and the cathode structure was collapsed, thus contributing to the leaching of Li. The proposed environmentally friendly recovery process of Li from spent NCM cathodes is promising for practical applications, offering significant economic benefits.
利用氧化和选择性浸出工艺从废旧锂离子电池中回收对环境友好的 Li2CO3
从废镍钴锰氧化物(NCM)阴极中提取有价金属通常需要使用强酸或强碱,这往往会导致二次污染。本文提出了一种以过硫酸钠(Na2S2O8)为唯一浸出剂选择性提取锂(Li)的环境友好型回收路线。在优化条件下,锂的浸出效率达到 98.02%,锂的选择性浸出效率为 94.80%。此外,从富锂滤液中回收的碳酸锂(Li2CO3)产品纯度高达 99.5%。湿化学、动力学、热力学和固相分析揭示了锂选择性浸出的机理。在选择性浸出过程中,Na2S2O8 产生了自由基 SO4-- 和 -OH、氢离子 (H+) 和钠离子 (Na+)。这些自由基可提高浸出系统的氧化还原电位。在这些条件下,Co 和 Mn 元素都保持在高价态,阴极结构坍塌,从而促进了锂的浸出。从废旧 NCM 阴极中回收锂的环境友好型工艺具有良好的实际应用前景,可带来显著的经济效益。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS ES&T engineering
ENGINEERING, ENVIRONMENTAL-
CiteScore
8.50
自引率
0.00%
发文量
0
期刊介绍:
ACS ES&T Engineering publishes impactful research and review articles across all realms of environmental technology and engineering, employing a rigorous peer-review process. As a specialized journal, it aims to provide an international platform for research and innovation, inviting contributions on materials technologies, processes, data analytics, and engineering systems that can effectively manage, protect, and remediate air, water, and soil quality, as well as treat wastes and recover resources.
The journal encourages research that supports informed decision-making within complex engineered systems and is grounded in mechanistic science and analytics, describing intricate environmental engineering systems. It considers papers presenting novel advancements, spanning from laboratory discovery to field-based application. However, case or demonstration studies lacking significant scientific advancements and technological innovations are not within its scope.
Contributions containing experimental and/or theoretical methods, rooted in engineering principles and integrated with knowledge from other disciplines, are welcomed.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


