Synthesis of hollow micro-mesoporous nitrogen-doped carbon nanoparticles for enhanced CO2 capture
Abstract
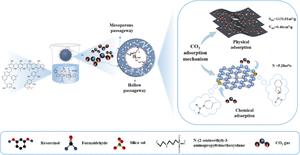
The design and synthesis of porous carbons with unique structures and diverse functionalities as CO2 adsorbents constitute a challenging and intriguing research topic. In this study, the synthesis of hollow micro-mesoporous nitrogen-doped carbon nanoparticles (NPCS) and its adsorption of CO2 were investigated. Highly porous nitrogen-doped carbon nanoparticles were successfully synthesized by using economically available resorcinol and formaldehyde as carbon precursors, with N-(2-aminoethyl)-3-aminopropyltrimethoxysilane (KH-792) as a soft template and silica sol as a hard template. The hollow nitrogen-doped carbon nanoparticles exhibit an evident microporous-mesoporous structure and have two different scales of mesopores with 9 nm and 12 nm, respectively. The effects of various synthetic parameters on the formation of hollow nitrogen-doped carbon nanoparticles were analyzed. The hollow nitrogen-doped carbon nanoparticles exhibited specific surface area of 1090 to 1716 m2/g and nitrogen content of 2.83 to 5.28%. At 273 K and 1 bar, the experimental results demonstrated the positive effects of the enriched pore structure and nitrogen doping on CO2 adsorption. The optimum adsorption capacity of activated NPCS (ANPCS) was 5.11 mmol/g with excellent CO2/N2 selectivity value of 20.44 at 273 K and 1 bar. The initial heat of adsorption value for ANPCS was 30.90 KJ/mol. Additionally, the hollow nitrogen-doped carbon nanoparticles retained 99.2% of the initial adsorbed amount after 5 cycles of adsorption. The excellent adsorption performance of the material can be ascribed not only to its extensive specific surface area and enriched nitrogen but also to its mesoporous and hollow structure, which facilitates rapid CO2 transport.
Graphical Abstract


 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


