PLD1 is a key player in cancer stemness and chemoresistance: Therapeutic targeting of cross-talk between the PI3K/Akt and Wnt/β-catenin pathways
IF 9.5
2区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
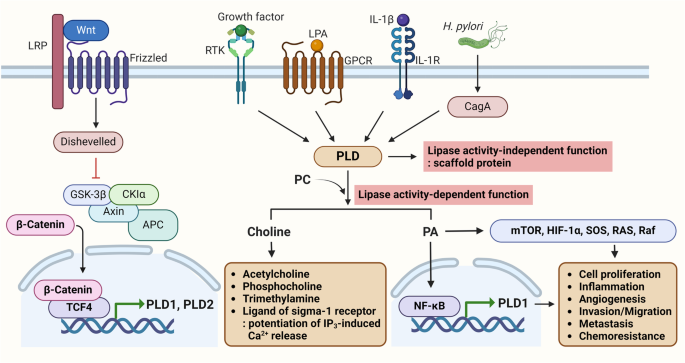
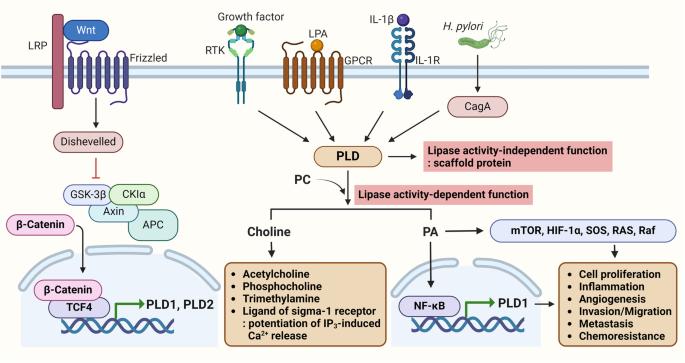
The development of chemoresistance is a major challenge in the treatment of several types of cancers in clinical settings. Stemness and chemoresistance are the chief causes of poor clinical outcomes. In this context, we hypothesized that understanding the signaling pathways responsible for chemoresistance in cancers is crucial for the development of novel targeted therapies to overcome drug resistance. Among the aberrantly activated pathways, the PI3K-Akt/Wnt/β-catenin signaling pathway is clinically implicated in malignancies such as colorectal cancer (CRC) and glioblastoma multiforme (GBM). Aberrant dysregulation of phospholipase D (PLD) has been implicated in several malignancies, and oncogenic activation of this pathway facilitates tumor proliferation, stemness, and chemoresistance. Crosstalk involving the PLD and Wnt/β-catenin pathways promotes the progression of CRC and GBM and reduces the sensitivity of cancer cells to standard therapies. Notably, both pathways are tightly regulated and connected at multiple levels by upstream and downstream effectors. Thus, gaining deeper insights into the interactions between these pathways would help researchers discover unique therapeutic targets for the management of drug-resistant cancers. Here, we review the molecular mechanisms by which PLD signaling stimulates stemness and chemoresistance in CRC and GBM. Thus, the current review aims to address the importance of PLD as a central player coordinating cross-talk between the PI3K/Akt and Wnt/β-catenin pathways and proposes the possibility of targeting these pathways to improve cancer therapy and overcome drug resistance. Cancer coming back after it seemed to have gone away is a big problem in treating cancers like colorectal cancer and a brain cancer called glioblastoma multiforme. This research looks at the part played by cancer stem cells (CSCs - cells within a tumor that can self-renew and cause the cancer to grow and come back) in cancer coming back and not responding to treatment. The scientists found that a pathway in the cells, called the Wnt/β-catenin signaling pathway, is important for keeping CSCs going. They also found that an enzyme (a type of protein that speeds up reactions in the body) called phospholipase D1 (PLD1) helps control this pathway. By stopping PLD1, they could lower the ability of CSCs to keep renewing themselves and make them more responsive to chemotherapy. This means that focusing on PLD1 could be a new way to treat cancers that don’t respond to existing treatments. This summary was initially drafted using artificial intelligence, then revised and fact-checked by the author.
PLD1 是癌症干性和化疗耐药性的关键角色以 PI3K/Akt 和 Wnt/β-catenin 通路之间的交叉对话为治疗目标。
化疗耐药性的产生是临床治疗多种癌症时面临的一大挑战。干细胞和化疗耐药性是导致临床疗效不佳的主要原因。在此背景下,我们假设,了解导致癌症化疗耐药性的信号通路对于开发新型靶向疗法以克服耐药性至关重要。在异常激活的通路中,PI3K-Akt/Wnt/β-catenin 信号通路与结直肠癌(CRC)和多形性胶质母细胞瘤(GBM)等恶性肿瘤有临床关联。磷脂酶 D(PLD)的异常失调与多种恶性肿瘤有关,该通路的致癌激活促进了肿瘤的增殖、干性和化疗抗性。涉及 PLD 和 Wnt/β-catenin 通路的串扰促进了 CRC 和 GBM 的进展,并降低了癌细胞对标准疗法的敏感性。值得注意的是,这两种通路都受到上游和下游效应物的严格调控,并在多个水平上相互连接。因此,深入了解这些通路之间的相互作用将有助于研究人员发现治疗耐药性癌症的独特靶点。在此,我们综述了PLD信号刺激CRC和GBM的干性和化疗耐药性的分子机制。因此,本综述旨在探讨PLD作为协调PI3K/Akt和Wnt/β-catenin通路之间交叉对话的核心参与者的重要性,并提出针对这些通路改善癌症治疗和克服耐药性的可能性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Experimental and Molecular Medicine
医学-生化与分子生物学
CiteScore
19.50
自引率
0.80%
发文量
166
审稿时长
3 months
期刊介绍:
Experimental & Molecular Medicine (EMM) stands as Korea's pioneering biochemistry journal, established in 1964 and rejuvenated in 1996 as an Open Access, fully peer-reviewed international journal. Dedicated to advancing translational research and showcasing recent breakthroughs in the biomedical realm, EMM invites submissions encompassing genetic, molecular, and cellular studies of human physiology and diseases. Emphasizing the correlation between experimental and translational research and enhanced clinical benefits, the journal actively encourages contributions employing specific molecular tools. Welcoming studies that bridge basic discoveries with clinical relevance, alongside articles demonstrating clear in vivo significance and novelty, Experimental & Molecular Medicine proudly serves as an open-access, online-only repository of cutting-edge medical research.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


