Understanding the Critical Bulk Properties of Zn-Salt Solution Electrolytes for Aqueous Zn-Ion Batteries
IF 7.2
2区 材料科学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
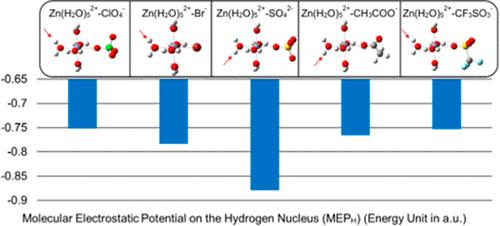
The unique technical merits of aqueous zinc-ion batteries (AZIBs) have attracted significant interest in the development of grid-scale energy storage technologies in the past decade. However, the development of AZIBs is severely hampered by the poor cycle stability, which exclusively stems from the electrolyte/electrode interactions. To address this issue, knowledge of the bulk properties of electrolytes, a pivotal component of AZIBs, is needed. Unfortunately, there still exists a significant gap in the data and understanding of these properties. This study investigates the concentration-dependent bulk properties of Zn-salt solution electrolytes through a combined experimental and theoretical approach. Key bulk properties such as pH, conductivity, water activity, hydrogen bonding, and electrochemical stability of five Zn-salt solutions are systematically studied as a function of concentration through a suite of experiments and theoretically interpreted by quantum chemistry calculations, molecular dynamics, and a tailored solvation model considering multispecies ion–ion and ion–molecule interactions. The model-produced theoretical results agree well with the experimental data. The revealed theoretical insights offer valuable fundamental guidance for future electrolyte discovery and understanding/mitigating degradation mechanisms in AZIBs.
了解用于锌-离子水电池的锌-盐溶液电解质的关键块体特性
在过去十年中,锌离子水电池(AZIBs)的独特技术优势引起了人们对电网级储能技术发展的极大兴趣。然而,循环稳定性差严重阻碍了 AZIB 的发展,而这完全源于电解质/电极之间的相互作用。为解决这一问题,需要了解作为 AZIBs 重要组成部分的电解质的体质特性。遗憾的是,在数据和对这些特性的理解方面仍存在很大差距。本研究通过实验和理论相结合的方法,研究了锌盐溶液电解质随浓度变化的体质特性。通过一系列实验,系统地研究了五种锌盐溶液的 pH 值、电导率、水活度、氢键和电化学稳定性等关键体态特性与浓度的函数关系,并通过量子化学计算、分子动力学和考虑多物种离子-离子和离子-分子相互作用的定制溶解模型进行了理论解释。模型得出的理论结果与实验数据非常吻合。所揭示的理论见解为未来发现电解质和理解/缓解 AZIB 的降解机制提供了宝贵的基础指导。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemistry of Materials
工程技术-材料科学:综合
CiteScore
14.10
自引率
5.80%
发文量
929
审稿时长
1.5 months
期刊介绍:
The journal Chemistry of Materials focuses on publishing original research at the intersection of materials science and chemistry. The studies published in the journal involve chemistry as a prominent component and explore topics such as the design, synthesis, characterization, processing, understanding, and application of functional or potentially functional materials. The journal covers various areas of interest, including inorganic and organic solid-state chemistry, nanomaterials, biomaterials, thin films and polymers, and composite/hybrid materials. The journal particularly seeks papers that highlight the creation or development of innovative materials with novel optical, electrical, magnetic, catalytic, or mechanical properties. It is essential that manuscripts on these topics have a primary focus on the chemistry of materials and represent a significant advancement compared to prior research. Before external reviews are sought, submitted manuscripts undergo a review process by a minimum of two editors to ensure their appropriateness for the journal and the presence of sufficient evidence of a significant advance that will be of broad interest to the materials chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


