Handy preparation of a carbon-Ni/NiO/Ni(OH)2 composite and its application in high-performance supercapacitors
Abstract
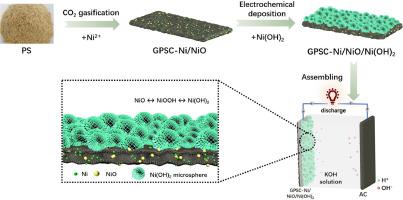
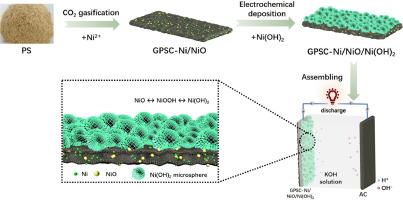
The growing demand for energy storage is required with the development of the intermittent renewable energy. Supercapacitors are a promising energy storage equipment, but their capacitive performance mainly depend on the electrode material. To obtain a high-performance electrode material for supercapacitors, in this work, a carbon-Ni/NiO/Ni(OH)2 composite was prepared from pine sawdust as the carbon precursor by integrating CO2 gasification and electrochemical deposition. It is found that the carbon-ternary nickel composite shows good electrochemical performance due to synergistic effect of the unique hierarchical structure and components involving porous carbon skeleton, Ni0 and NiO nanoparticles, and Ni(OH)2 microspheres. The composite displays the specific capacitance of 1875.6 F/g (or 937.8 C/g) at a current density of 1 A/g in a traditional three-electrode system. The assembled asymmetric supercapacitor device presents a potential window of 1.6 V, along with the energy density of 38.24 Wh/kg at the power density of 400 W/kg (or 22.60 Wh/kg at 2000 W/kg). After 6000 charge-discharge cycles, the capacitance retention ratio of the assembled supercapacitor reaches up to 105 %, exhibiting a good application potential. This work provides a novel and handy strategy for preparation of ternary nickel-based composite for advanced supercapacitors.

| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


