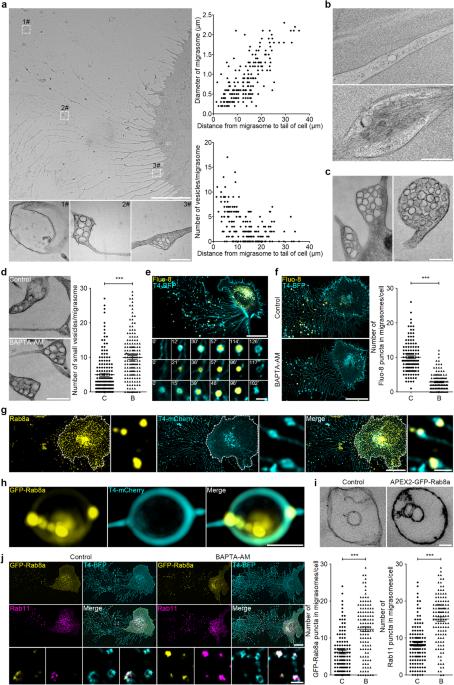
Localized, highly efficient secretion of signaling proteins by migrasomes
IF 28.1
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
Migrasomes, enriched with signaling molecules such as chemokines, cytokines and angiogenic factors, play a pivotal role in the spatially defined delivery of these molecules, influencing critical physiological processes including organ morphogenesis and angiogenesis. The mechanism governing the accumulation of signaling molecules in migrasomes has been elusive. In this study, we show that secretory proteins, including signaling proteins, are transported into migrasomes by secretory carriers via both the constitutive and regulated secretion pathways. During cell migration, a substantial portion of these carriers is redirected to the rear of the cell and actively transported into migrasomes, driven by the actin-dependent motor protein Myosin-5a. Once at the migrasomes, these carriers fuse with the migrasome membrane through SNARE-mediated mechanisms. Inhibiting migrasome formation significantly reduces secretion, suggesting migrasomes as a principal secretion route in migrating cells. Our findings reveal a specialized, highly localized secretion paradigm in migrating cells, conceptually paralleling the targeted neurotransmitter release observed in neuronal systems.

迁移体局部高效分泌信号蛋白
移行体富含趋化因子、细胞因子和血管生成因子等信号分子,在这些分子的空间传递中发挥着关键作用,影响着器官形态发生和血管生成等关键生理过程。信号分子在移行体中的积累机制一直难以捉摸。在这项研究中,我们发现分泌蛋白(包括信号蛋白)通过组成型分泌途径和调节型分泌途径被分泌载体运输到迁移体中。在细胞迁移过程中,这些载体的很大一部分被重新定向到细胞后部,并在肌动蛋白依赖性马达蛋白肌球蛋白-5a的驱动下被主动运输到迁移体中。一旦进入移行体,这些载体就会通过 SNARE 介导的机制与移行体膜融合。抑制迁移体的形成可显著减少分泌,这表明迁移体是迁移细胞的主要分泌途径。我们的研究结果揭示了迁移细胞中一种专门的、高度局部化的分泌模式,在概念上与神经元系统中观察到的定向神经递质释放相似。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Cell Research
生物-细胞生物学
CiteScore
53.90
自引率
0.70%
发文量
2420
审稿时长
2.3 months
期刊介绍:
Cell Research (CR) is an international journal published by Springer Nature in partnership with the Center for Excellence in Molecular Cell Science, Chinese Academy of Sciences (CAS). It focuses on publishing original research articles and reviews in various areas of life sciences, particularly those related to molecular and cell biology. The journal covers a broad range of topics including cell growth, differentiation, and apoptosis; signal transduction; stem cell biology and development; chromatin, epigenetics, and transcription; RNA biology; structural and molecular biology; cancer biology and metabolism; immunity and molecular pathogenesis; molecular and cellular neuroscience; plant molecular and cell biology; and omics, system biology, and synthetic biology. CR is recognized as China's best international journal in life sciences and is part of Springer Nature's prestigious family of Molecular Cell Biology journals.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


