High internal phase emulsions gels stabilized by soy protein isolate and rutin complexes: Encapsulation, interfacial properties and in vitro digestibility
Abstract
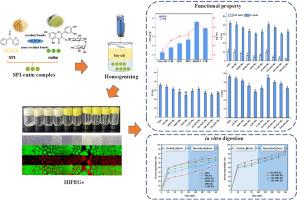
High internal phase emulsion gel (HIPEG) stabilized by food-grade protein particles has garnered increasing attention across various fields. However, the limited environmental stability in HIPEGs remain key challenges. In this study, oil-in-water HIPEGs stabilized by soy protein isolate and rutin complexes was used as a model system. The impacts of different conjugation conditions in preparing SPI and rutin (SPI-R) complex and various homogenization rates on HIPEGs formation were investigated. Results demonstrated that HIPEGs stabilized by covalent binding SPI-R complex exhibited excellent interfacial properties with more proteins involved in the formation of the HIPEGs at the interface compared to noncovalent-conjugate-stabilized HIPEGs. Notably, HIPEGs stabilized by SPI-R complex under alkaline conditions showed narrower size distribution and a more compact droplet structure along with a denser gel network and enhanced encapsulation efficiency of rutin compared to other groups. Moreover, the HIPEGs stabilized by these covalent conjugates exhibited better environmental stability and superior digestibility of protein. Specifically, HIPEGs stabilized by alkaline-conditioned SPI-R complex demonstrated excellent kinetically or thermodynamically stable state with minimal loss of stability under different environmental stresses when homogenized at an optimal rate of 15,000 rpm/min. This study provides valuable insights into utilizing protein-polyphenol particles as outstanding emulsifiers for food-grade HIPEGs.

| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


