Quantitative control of subcellular protein localization with a photochromic dimerizer
IF 12.9
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Artificial control of intracellular protein dynamics with high precision provides deep insight into complicated biomolecular networks. Optogenetics and caged compound-based chemically induced dimerization (CID) systems are emerging as tools for spatiotemporally regulating intracellular protein dynamics. However, both technologies face several challenges for accurate control such as the duration of activation, deactivation rate and repetition cycles. Herein, we report a photochromic CID system that uses the photoisomerization of a ligand so that both association and dissociation are controlled by light, enabling quick, repetitive and quantitative regulation of the target protein localization upon illumination with violet and green light. We also demonstrate the usability of the photochromic CID system as a potential tool to finely manipulate intracellular protein dynamics during multicolor fluorescence imaging to study diverse cellular processes. We use this system to manipulate PTEN-induced kinase 1 (PINK1)–Parkin-mediated mitophagy, showing that PINK1 recruitment to the mitochondria can promote Parkin recruitment to proceed with mitophagy. A photochromic dimerizer was developed for light-controlled reversible and quantitative regulation of intracellular protein localization, enabling optical control of PTEN-induced kinase 1 (PINK1)–Parkin-mediated mitophage induction.
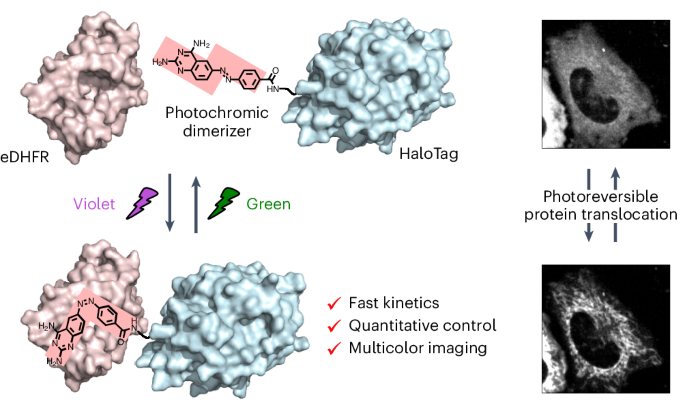
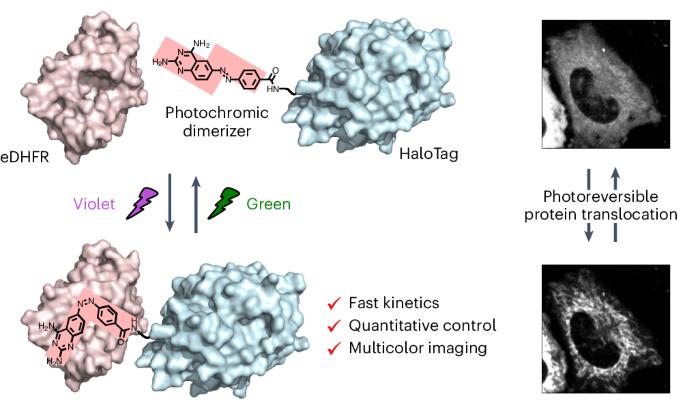
用光变色二聚体定量控制亚细胞蛋白质定位
对细胞内蛋白质动态进行高精度的人工控制,有助于深入了解复杂的生物分子网络。光遗传学和基于笼状化合物的化学诱导二聚化(CID)系统正在成为时空调控细胞内蛋白质动态的工具。然而,这两种技术在精确控制方面都面临着一些挑战,如激活持续时间、失活率和重复周期。在本文中,我们报告了一种光致变色 CID 系统,该系统利用配体的光异构化,使其结合和解离均由光控制,从而在紫光和绿光照射下快速、重复和定量地调节目标蛋白质的定位。我们还展示了光致变色 CID 系统的可用性,它是一种在多色荧光成像过程中精细操纵细胞内蛋白质动态的潜在工具,可用于研究各种细胞过程。我们利用该系统操纵 PTEN 诱导的激酶 1 (PINK1)-Parkin 介导的有丝分裂,结果表明 PINK1 招募到线粒体可以促进 Parkin 招募,从而进行有丝分裂。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature chemical biology
生物-生化与分子生物学
CiteScore
23.90
自引率
1.40%
发文量
238
审稿时长
12 months
期刊介绍:
Nature Chemical Biology stands as an esteemed international monthly journal, offering a prominent platform for the chemical biology community to showcase top-tier original research and commentary. Operating at the crossroads of chemistry, biology, and related disciplines, chemical biology utilizes scientific ideas and approaches to comprehend and manipulate biological systems with molecular precision.
The journal embraces contributions from the growing community of chemical biologists, encompassing insights from chemists applying principles and tools to biological inquiries and biologists striving to comprehend and control molecular-level biological processes. We prioritize studies unveiling significant conceptual or practical advancements in areas where chemistry and biology intersect, emphasizing basic research, especially those reporting novel chemical or biological tools and offering profound molecular-level insights into underlying biological mechanisms.
Nature Chemical Biology also welcomes manuscripts describing applied molecular studies at the chemistry-biology interface due to the broad utility of chemical biology approaches in manipulating or engineering biological systems. Irrespective of scientific focus, we actively seek submissions that creatively blend chemistry and biology, particularly those providing substantial conceptual or methodological breakthroughs with the potential to open innovative research avenues. The journal maintains a robust and impartial review process, emphasizing thorough chemical and biological characterization.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


