Synthesis and physicochemical evaluation of tetrafluorinated GABA analogue
IF 1.7
4区 化学
Q3 CHEMISTRY, INORGANIC & NUCLEAR
引用次数: 0
Abstract
Synthesis of 2,2,3,3-tetraftluoro-γ-aminobutyric acid (tetraF-GABA) starting from commercially available 2,2,3,3-tetrafluorobutane-1,4-diol is described. The straightforward nine-step reaction sequence allowed the production of up to 20 g of the target amino acid in 41 % overall yield. Potentiometric titration revealed increased acidity of both ionizable moieties in the title compound (the pKa values were diminished by 1.6 and 4.0 units for the COOH and NH3+ groups, respectively, as compared to parent GABA). X-ray diffraction studies and DFT calculations demonstrated different preferential conformations in solid state and solution, although the fluorine gauche effect was observed in both cases.
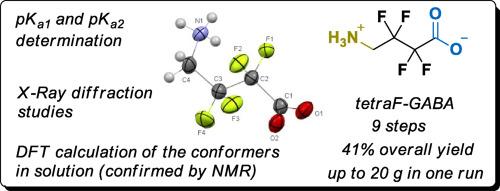
四氟 GABA 类似物的合成与理化评价
本文介绍了从市售的 2,2,3,3- 四氟丁烷-1,4-二醇开始合成 2,2,3,3-四氟-γ-氨基丁酸(tetraF-GABA)的过程。通过简单的九步反应顺序,可以生产出最多 20 克的目标氨基酸,总收率为 41%。电位滴定显示,标题化合物中两个可电离分子的酸性都有所增强(与母体 GABA 相比,COOH 和 NH3+ 基团的 pKa 值分别降低了 1.6 和 4.0 个单位)。X 射线衍射研究和 DFT 计算表明,尽管在两种情况下都观察到了氟高切效应,但固态和溶液中的优先构象有所不同。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Fluorine Chemistry
化学-无机化学与核化学
CiteScore
3.80
自引率
10.50%
发文量
99
审稿时长
33 days
期刊介绍:
The Journal of Fluorine Chemistry contains reviews, original papers and short communications. The journal covers all aspects of pure and applied research on the chemistry as well as on the applications of fluorine, and of compounds or materials where fluorine exercises significant effects. This can include all chemistry research areas (inorganic, organic, organometallic, macromolecular and physical chemistry) but also includes papers on biological/biochemical related aspects of Fluorine chemistry as well as medicinal, agrochemical and pharmacological research. The Journal of Fluorine Chemistry also publishes environmental and industrial papers dealing with aspects of Fluorine chemistry on energy and material sciences. Preparative and physico-chemical investigations as well as theoretical, structural and mechanistic aspects are covered. The Journal, however, does not accept work of purely routine nature.
For reviews and special issues on particular topics of fluorine chemistry or from selected symposia, please contact the Regional Editors for further details.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


