A nucleophilic beryllyl complex via metathesis at [Be–Be]2+
IF 19.2
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
Owing to its high toxicity, the chemistry of element number four, beryllium, is poorly understood. However, as the lightest elements provide the basis for fundamental models of chemical bonding, there is a need for greater insight into the properties of beryllium. In this context, the chemistry of the homo-elemental Be–Be bond is of fundamental interest. Here the ligand metathesis chemistry of diberyllocene (1; CpBeBeCp)—a stable complex with a Be–Be bond—has been investigated. These studies yield two complexes with Be–Be bonds: Cp*BeBeCp (2) and [K{(HCDippN)2BO}2]BeBeCp (3; Dipp = 2,6-diisopropylphenyl). Quantum chemical calculations indicate that the Be–Be bond in 3 is polarized to such an extent that the complex could be formulated as a mixed-oxidation state Be0/BeII complex. Correspondingly, it is demonstrated that 3 can transfer the ‘beryllyl’ anion, [BeCp]−, to an organic substrate, by analogy with the reactivity of sp2–sp3 diboranes. Indeed, this work reveals striking similarities between the homo-elemental bonding linkages of beryllium and boron, despite the respective metallic and non-metallic natures of these elements. The chemistry of beryllium, one of the periodic table’s lightest elements, remains poorly understood. Now, a nucleophilic beryllium complex with a highly polarized Be–Be bond has been prepared—this compound could be seen as a mixed-oxidation state Be(0)/Be(II) complex. The Be–Be bond is cleaved upon reaction with an organic substrate.
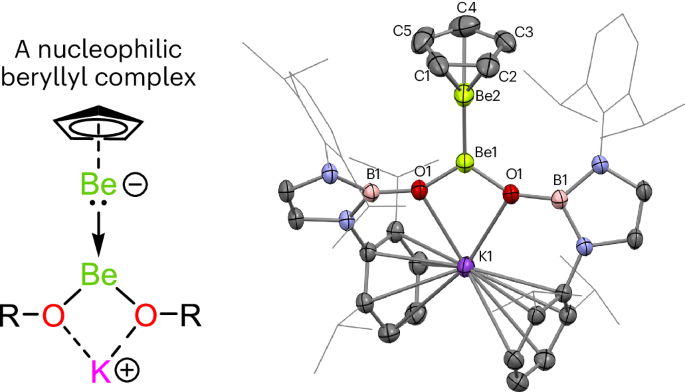
通过[Be-Be]2+的偏析作用生成亲核伯利基络合物
由于毒性很强,人们对四号元素铍的化学性质知之甚少。然而,由于最轻的元素为化学键的基本模型提供了基础,因此需要更深入地了解铍的特性。在这方面,同元素铍-铍键的化学性质具有重要意义。在此,我们研究了具有 Be-Be 键的稳定络合物--二赤藓烯(1;CpBeBeCp)的配体偏合成化学。这些研究产生了两种具有 Be-Be 键的配合物:Cp*BeBeCp (2) 和 [K{(HCDippN)2BO}2]BeBeCp (3; Dipp = 2,6-二异丙基苯基)。量子化学计算表明,3 中的 Be-Be 键被极化,以至于该复合物可以被表述为混合氧化态 Be0/BeII 复合物。相应地,通过类比 sp2-sp3 二硼烷的反应性,证明 3 可以将 "伯利 "阴离子 [BeCp]- 转移到有机底物上。事实上,尽管铍和硼分别具有金属和非金属的性质,但这项研究揭示了这两种元素的同元素键联之间惊人的相似性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature chemistry
化学-化学综合
CiteScore
29.60
自引率
1.40%
发文量
226
审稿时长
1.7 months
期刊介绍:
Nature Chemistry is a monthly journal that publishes groundbreaking and significant research in all areas of chemistry. It covers traditional subjects such as analytical, inorganic, organic, and physical chemistry, as well as a wide range of other topics including catalysis, computational and theoretical chemistry, and environmental chemistry.
The journal also features interdisciplinary research at the interface of chemistry with biology, materials science, nanotechnology, and physics. Manuscripts detailing such multidisciplinary work are encouraged, as long as the central theme pertains to chemistry.
Aside from primary research, Nature Chemistry publishes review articles, news and views, research highlights from other journals, commentaries, book reviews, correspondence, and analysis of the broader chemical landscape. It also addresses crucial issues related to education, funding, policy, intellectual property, and the societal impact of chemistry.
Nature Chemistry is dedicated to ensuring the highest standards of original research through a fair and rigorous review process. It offers authors maximum visibility for their papers, access to a broad readership, exceptional copy editing and production standards, rapid publication, and independence from academic societies and other vested interests.
Overall, Nature Chemistry aims to be the authoritative voice of the global chemical community.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


