Vimentin filaments integrate low-complexity domains in a complex helical structure
IF 12.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Intermediate filaments (IFs) are integral components of the cytoskeleton. They provide cells with tissue-specific mechanical properties and are involved in numerous cellular processes. Due to their intricate architecture, a 3D structure of IFs has remained elusive. Here we use cryo-focused ion-beam milling, cryo-electron microscopy and tomography to obtain a 3D structure of vimentin IFs (VIFs). VIFs assemble into a modular, intertwined and flexible helical structure of 40 α-helices in cross-section, organized into five protofibrils. Surprisingly, the intrinsically disordered head domains form a fiber in the lumen of VIFs, while the intrinsically disordered tails form lateral connections between the protofibrils. Our findings demonstrate how protein domains of low sequence complexity can complement well-folded protein domains to construct a biopolymer with striking mechanical strength and stretchability. Using cryo-electron microscopy and integrative modeling, the authors defined the structure of vimentin intermediate filaments, revealing a helical tube built of five protofibrils that enclose a fiber of low-complexity N-terminal domains.
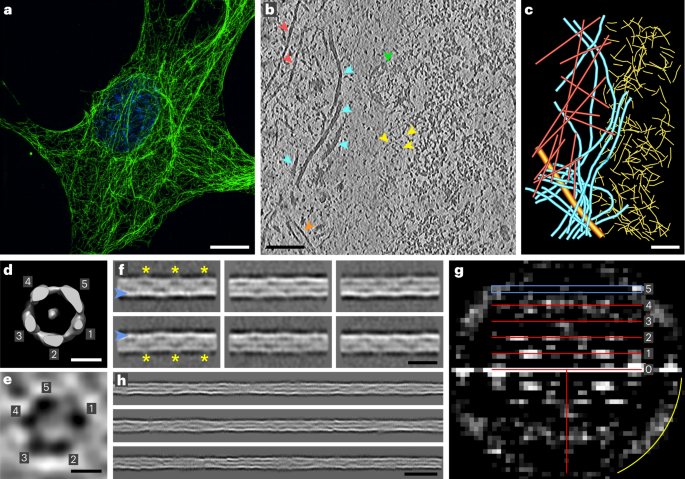
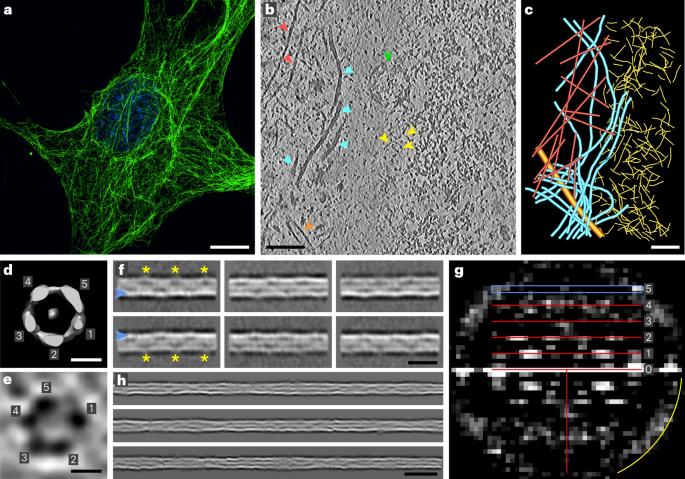
波形蛋白丝在复杂的螺旋结构中整合了低复杂度结构域
中间丝(IFs)是细胞骨架不可或缺的组成部分。它们为细胞提供组织特异性的机械特性,并参与许多细胞过程。由于其结构复杂,中间丝的三维结构一直难以捉摸。在这里,我们利用低温聚焦离子束铣削、低温电子显微镜和层析成像技术获得了波形蛋白IFs(VIFs)的三维结构。VIFs 由横截面为 40 α-螺旋的模块化、交织和灵活的螺旋结构组成,并组织成五条原纤维。令人惊讶的是,内在无序的头部结构域在 VIF 的内腔中形成纤维,而内在无序的尾部结构域则在原纤维之间形成横向连接。我们的研究结果表明,低序列复杂性的蛋白质结构域可以与折叠良好的蛋白质结构域互补,从而构建出具有惊人机械强度和拉伸性的生物聚合物。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Structural & Molecular Biology
BIOCHEMISTRY & MOLECULAR BIOLOGY-BIOPHYSICS
CiteScore
22.00
自引率
1.80%
发文量
160
审稿时长
3-8 weeks
期刊介绍:
Nature Structural & Molecular Biology is a comprehensive platform that combines structural and molecular research. Our journal focuses on exploring the functional and mechanistic aspects of biological processes, emphasizing how molecular components collaborate to achieve a particular function. While structural data can shed light on these insights, our publication does not require them as a prerequisite.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


