Dinuclear Cu(I) molecular electrocatalyst for CO2-to-C3 product conversion
IF 42.8
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
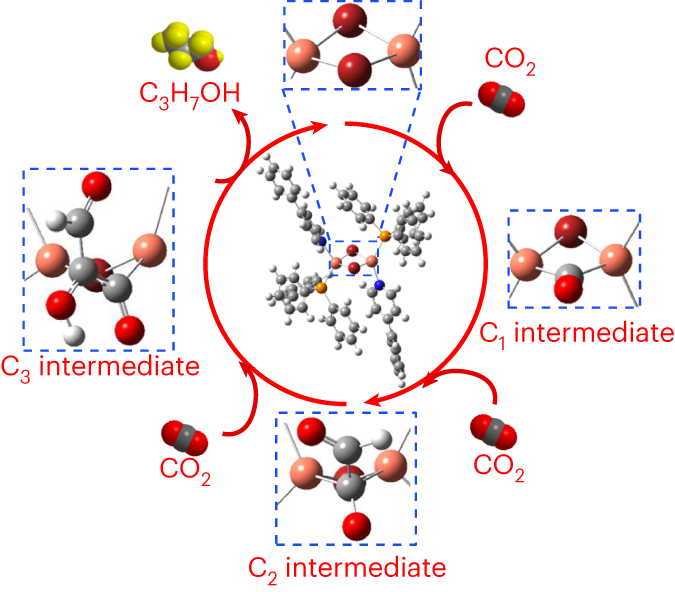
Molecular metal complex catalysts are highly tunable in terms of their CO2 reduction performance by means of their flexible molecular design. However, metal complex catalysts have challenges in their structural stability and it has not been possible to synthesize high-value-added C3 products due to their inability to perform C–C coupling. Here we show a CO2 reduction reaction catalysed by a Br-bridged dinuclear Cu(I) complex that produces C3H7OH with high robustness during the reaction. The C–C coupling reaction mechanism was analysed by experimental operando surface-enhanced Raman scattering analysis, and theoretical quantum-chemical calculations proposed the formation of a C–C coupling intermediate species with substrate incorporation between the two Cu centres. Molecular design guidelines based on this discovery offer an approach to developing next-generation catalysts that generate multicarbon CO2 reduction products. The tunable design of molecular catalysts presents opportunities for the control of product selectivity in CO2 reduction, yet to date, complexes capable of producing multicarbon products have been elusive. Here, a Br-bridged dinuclear Cu(I) complex that turns over C3H7OH is reported.
将 CO2 转化为 C3 产物的双核 Cu(I)分子电催化剂
分子金属络合催化剂通过灵活的分子设计,可对其二氧化碳还原性能进行高度调整。然而,金属络合物催化剂在结构稳定性方面存在挑战,而且由于其无法进行 C-C 偶联,因此一直无法合成高附加值的 C3 产物。在此,我们展示了一种由桥桥双核 Cu(I) 复合物催化的 CO2 还原反应,该反应在反应过程中能产生 C3H7OH,且具有很高的稳定性。我们通过实验操作表面增强拉曼散射分析分析了 C-C 耦合反应机理,并通过量子化学理论计算提出了在两个 Cu 中心之间形成底物结合的 C-C 耦合中间产物。基于这一发现的分子设计指南为开发生成多碳二氧化碳还原产物的下一代催化剂提供了一种方法。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Catalysis
Chemical Engineering-Bioengineering
CiteScore
52.10
自引率
1.10%
发文量
140
期刊介绍:
Nature Catalysis serves as a platform for researchers across chemistry and related fields, focusing on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, encompassing both fundamental and applied studies. With a particular emphasis on advancing sustainable industries and processes, the journal provides comprehensive coverage of catalysis research, appealing to scientists, engineers, and researchers in academia and industry.
Maintaining the high standards of the Nature brand, Nature Catalysis boasts a dedicated team of professional editors, rigorous peer-review processes, and swift publication times, ensuring editorial independence and quality. The journal publishes work spanning heterogeneous catalysis, homogeneous catalysis, and biocatalysis, covering areas such as catalytic synthesis, mechanisms, characterization, computational studies, nanoparticle catalysis, electrocatalysis, photocatalysis, environmental catalysis, asymmetric catalysis, and various forms of organocatalysis.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


