Antiangiogenic–immune-checkpoint inhibitor combinations: lessons from phase III clinical trials
IF 81.1
1区 医学
Q1 ONCOLOGY
引用次数: 0
Abstract
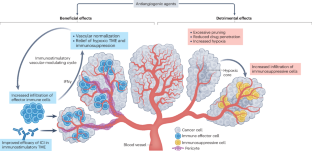
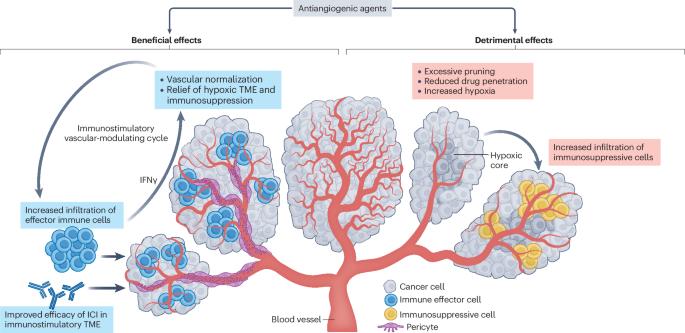
Antiangiogenic agents, generally antibodies or tyrosine-kinase inhibitors that target the VEGF–VEGFR pathway, are currently among the few combination partners clinically proven to improve the efficacy of immune-checkpoint inhibitors (ICIs). This benefit has been demonstrated in pivotal phase III trials across different cancer types, some with practice-changing results; however, numerous phase III trials have also had negative results. The rationale for using antiangiogenic drugs as partners for ICIs relies primarily on blocking the multiple immunosuppressive effects of VEGF and inducing several different vascular-modulating effects that can stimulate immunity, such as vascular normalization leading to increased intratumoural blood perfusion and flow, and inhibition of pro-apoptotic effects of endothelial cells on T cells, among others. Conversely, VEGF blockade can also cause changes that suppress antitumour immunity, such as increased tumour hypoxia, and reduced intratumoural ingress of co-administered ICIs. As a result, the net clinical benefits from antiangiogenic–ICI combinations will be determined by the balance between the opposing effects of VEGF signalling and its inhibition on the antitumour immune response. In this Perspective, we summarize the results from the currently completed phase III trials evaluating antiangiogenic agent–ICI combinations. We also discuss strategies to improve the efficacy of these combinations, focusing on aspects that include the deleterious functions of VEGF–VEGFR inhibition on antitumour immunity, vessel co-option as a driver of non-angiogenic tumour growth, clinical trial design, or the rationale for drug selection, dosing and scheduling. The benefit of combining antiangiogenic agents with immune-checkpoint inhibitors has been demonstrated in pivotal phase III trials across different cancer types, some with practice-changing results; however, other phase III trials have had negative results. The authors of this Perspective discuss the variable outcomes of these trials, considering factors that account for these differences and suggesting future initiatives for improving the outcomes in patients receiving these combinations.
抗血管生成-免疫检查点抑制剂组合:III 期临床试验的启示
抗血管生成药物(通常是针对血管内皮生长因子-血管内皮生长因子受体通路的抗体或酪氨酸激酶抑制剂)是目前少数几种经临床证实可提高免疫检查点抑制剂(ICIs)疗效的联合用药。这种益处已在不同癌症类型的关键性 III 期试验中得到证实,其中一些试验的结果改变了临床实践;然而,许多 III 期试验也出现了负面结果。使用抗血管生成药物作为 ICIs 伴侣的主要理由是阻断血管内皮生长因子的多种免疫抑制作用,并诱导多种不同的血管调节作用,从而刺激免疫,如血管正常化导致瘤内血液灌注和流动增加,以及抑制内皮细胞对 T 细胞的促凋亡作用等。相反,血管内皮生长因子阻断也会导致抑制抗肿瘤免疫的变化,如增加肿瘤缺氧、减少合用 ICIs 的瘤内摄取等。因此,抗血管生成-ICI 联合疗法的净临床获益将取决于血管内皮生长因子信号传递及其抑制对抗肿瘤免疫反应的对立效应之间的平衡。在本透视中,我们总结了目前已完成的抗血管生成剂-ICI 联合疗法 III 期临床试验的评估结果。我们还讨论了提高这些组合疗效的策略,重点关注的方面包括 VEGF-VEGFR 抑制对抗肿瘤免疫的有害作用、作为非血管生成性肿瘤生长驱动因素的血管增生、临床试验设计或药物选择、剂量和时间安排的合理性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
99.40
自引率
0.40%
发文量
114
审稿时长
6-12 weeks
期刊介绍:
Nature Reviews publishes clinical content authored by internationally renowned clinical academics and researchers, catering to readers in the medical sciences at postgraduate levels and beyond. Although targeted at practicing doctors, researchers, and academics within specific specialties, the aim is to ensure accessibility for readers across various medical disciplines. The journal features in-depth Reviews offering authoritative and current information, contextualizing topics within the history and development of a field. Perspectives, News & Views articles, and the Research Highlights section provide topical discussions, opinions, and filtered primary research from diverse medical journals.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


