Bacteria-mediated resistance of neutrophil extracellular traps to enzymatic degradation drives the formation of dental calculi
IF 26.8
1区 医学
Q1 ENGINEERING, BIOMEDICAL
引用次数: 0
Abstract
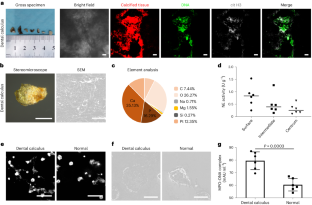
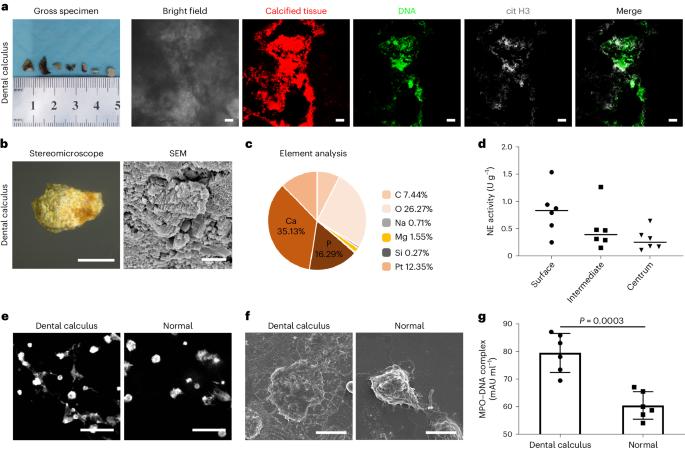
Dental calculi can cause gingival bleeding and periodontitis, yet the mechanism underlying the formation of such mineral build-ups, and in particular the role of the local microenvironment, are unclear. Here we show that the formation of dental calculi involves bacteria in local mature biofilms converting the DNA in neutrophil extracellular traps (NETs) from being degradable by the enzyme DNase I to being degradation resistant, promoting the nucleation and growth of apatite. DNase I inhibited NET-induced mineralization in vitro and ex vivo, yet plasma DNases were ineffective at inhibiting ectopic mineralization in the oral cavity in rodents. The topical application of the DNA-intercalating agent chloroquine in rodents fed with a dental calculogenic diet reverted NET DNA to its degradable form, inhibiting the formation of calculi. Our findings may motivate therapeutic strategies for the reduction of the prevalence of the deposition of bacteria-driven calculi in the oral cavity. The development of dental calculi involves bacteria in local mature biofilms converting the DNA in neutrophil extracellular traps from being degradable by the enzyme DNase I to being degradation resistant.
细菌介导的中性粒细胞胞外捕获物对酶降解的抵抗力促使牙结石的形成。
牙结石可导致牙龈出血和牙周炎,但这种矿物质堆积的形成机制,尤其是局部微环境的作用尚不清楚。在这里,我们发现牙结石的形成涉及局部成熟生物膜中的细菌将中性粒细胞胞外捕获物(NET)中的 DNA 从可被 DNase I 酶降解转化为耐降解,从而促进了磷灰石的成核和生长。DNase I 可抑制体外和体内由细胞外捕获物诱导的矿化,但血浆中的 DNase 无法有效抑制啮齿类动物口腔中的异位矿化。在啮齿类动物体内局部使用DNA拮抗剂氯喹可将NET DNA还原为可降解形式,从而抑制结石的形成。我们的研究结果可能有助于制定治疗策略,减少口腔中由细菌引起的结石沉积。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Biomedical Engineering
Medicine-Medicine (miscellaneous)
CiteScore
45.30
自引率
1.10%
发文量
138
期刊介绍:
Nature Biomedical Engineering is an online-only monthly journal that was launched in January 2017. It aims to publish original research, reviews, and commentary focusing on applied biomedicine and health technology. The journal targets a diverse audience, including life scientists who are involved in developing experimental or computational systems and methods to enhance our understanding of human physiology. It also covers biomedical researchers and engineers who are engaged in designing or optimizing therapies, assays, devices, or procedures for diagnosing or treating diseases. Additionally, clinicians, who make use of research outputs to evaluate patient health or administer therapy in various clinical settings and healthcare contexts, are also part of the target audience.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


