Interactions between a neutral polymeric bonding agent and nitramine explosives and their influencing factors
IF 3.9
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
Neutral polymer bonding agents (NPBAs) have proven highly effective in enhancing the interfacial bonding between the bonding matrix and nitramine explosives such as cyclic trimethylene trinitramine (RDX) and cyclic tetramethylene trinitramine (HMX). However, there is a lack of clear understanding of the mechanisms behind their interactions, and it has been found that NPBAs produce different interfacial bonding effects on RDX and HMX. To gain deeper insights into the molecular-scale interfacial interactions of nitramine explosives/NPBAs, this study investigated the molecular electrostatic potentials, intermolecular interactions, surface structural features, and interfacial adsorption of a NPBA onto nitramine explosives using the density functional theory and molecular dynamics (MD) technique. The results indicate that the N atom on the cyano group of the NPBA molecule can form weak hydrogen bonds C–H⋅⋅⋅N![]() C and C–H⋅⋅⋅O with the H atoms in the RDX and HMX molecules. The strength of such weak hydrogen bonding interactions is affected by the electrostatic potential range of nitramine molecules. Additionally, the surface structure of the nitramine plays a critical role in the NPBA adsorption strength. Compared to HMX, RDX exhibits a narrower surface electrostatic potential range and smoother crystal surface, resulting in weaker intermolecular interactions between the NPBA and the RDX surface.
C and C–H⋅⋅⋅O with the H atoms in the RDX and HMX molecules. The strength of such weak hydrogen bonding interactions is affected by the electrostatic potential range of nitramine molecules. Additionally, the surface structure of the nitramine plays a critical role in the NPBA adsorption strength. Compared to HMX, RDX exhibits a narrower surface electrostatic potential range and smoother crystal surface, resulting in weaker intermolecular interactions between the NPBA and the RDX surface.
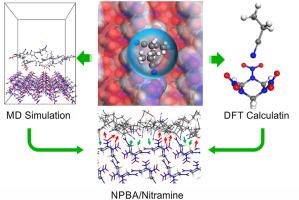
中性聚合粘接剂与硝胺炸药之间的相互作用及其影响因素
事实证明,中性聚合物粘接剂(NPBAs)在增强粘接基体与环三亚甲基三硝胺(RDX)和环四亚甲基三硝胺(HMX)等硝胺炸药之间的界面粘接方面非常有效。然而,人们对其相互作用背后的机制还缺乏清晰的认识,而且发现 NPBA 对 RDX 和 HMX 产生不同的界面键合效应。为了深入了解硝胺炸药/NPBA 的分子尺度界面相互作用,本研究利用密度泛函理论和分子动力学(MD)技术研究了 NPBA 在硝胺炸药上的分子静电势、分子间相互作用、表面结构特征以及界面吸附。结果表明,NPBA 分子氰基上的 N 原子可与 RDX 和 HMX 分子中的 H 原子形成 C-H⋅⋅⋅N C 和 C-H⋅⋅⋅O 弱氢键。这种弱氢键相互作用的强度受到硝胺分子静电电位范围的影响。此外,硝胺的表面结构对 NPBA 吸附强度也起着关键作用。与 HMX 相比,RDX 的表面静电势范围更窄,晶面更光滑,因此 NPBA 与 RDX 表面之间的分子间相互作用更弱。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Energetic Materials Frontiers
Materials Science-Materials Science (miscellaneous)
CiteScore
6.90
自引率
0.00%
发文量
42
审稿时长
12 weeks
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


