Targeting cuproplasia and cuproptosis in cancer
IF 82.2
1区 医学
Q1 ONCOLOGY
引用次数: 0
Abstract
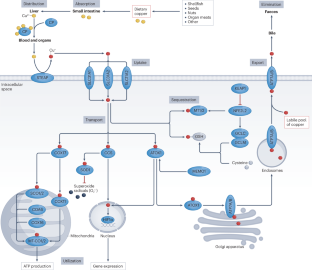
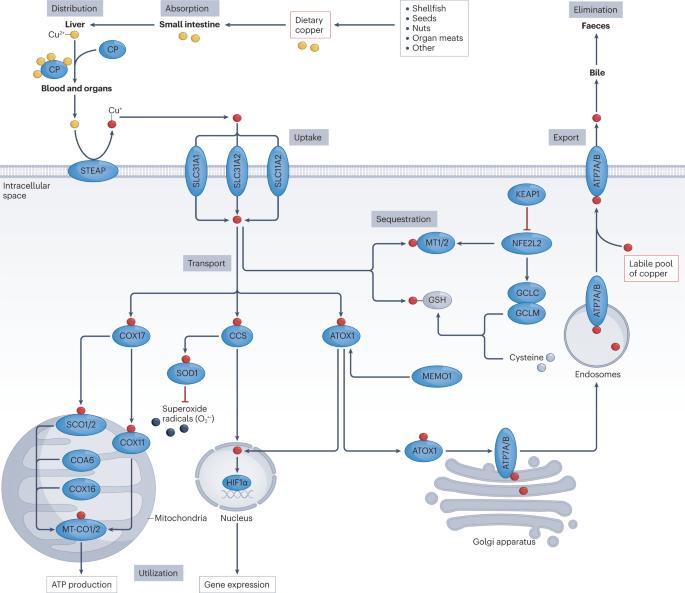
Copper, an essential trace element that exists in oxidized and reduced forms, has pivotal roles in a variety of biological processes, including redox chemistry, enzymatic reactions, mitochondrial respiration, iron metabolism, autophagy and immune modulation; maintaining copper homeostasis is crucial as both its deficiency and its excess are deleterious. Dysregulated copper metabolism has a dual role in tumorigenesis and cancer therapy. Specifically, cuproplasia describes copper-dependent cell growth and proliferation, including hyperplasia, metaplasia and neoplasia, whereas cuproptosis refers to a mitochondrial pathway of cell death triggered by excessive copper exposure and subsequent proteotoxic stress (although complex interactions between cuproptosis and other cell death mechanisms, such as ferroptosis, are likely and remain enigmatic). In this Review, we summarize advances in our understanding of copper metabolism, the molecular machineries underlying cuproplasia and cuproptosis, and their potential targeting for cancer therapy. These new findings advance the rapidly expanding field of translational cancer research focused on metal compounds. Copper is an essential trace element with inherent redox properties and fundamental roles in a diverse range of biological processes; therefore, maintaining copper homeostasis is crucial. In this Review, the authors discuss new insights into the mechanisms by which disrupted copper homeostasis contributes to tumour initiation and development, including the recently defined concepts of cuproplasia (copper-dependent cell growth and proliferation) and cuproptosis (a mitochondrial pathway of cell death triggered by excessive copper exposure). They also discuss potential strategies to exploit cuproplasia and cuproptosis for the treatment of cancer.
以癌症中的杯状增生和杯状突变为目标
铜是一种以氧化和还原形式存在的必需微量元素,在氧化还原化学、酶反应、线粒体呼吸、铁代谢、自噬和免疫调节等多种生物过程中发挥着关键作用。铜代谢失调在肿瘤发生和癌症治疗中具有双重作用。具体来说,铜增生是指依赖铜的细胞生长和增殖,包括增生、变性和肿瘤,而铜中毒则是指铜暴露过量和随后的蛋白毒性应激引发的线粒体细胞死亡途径(尽管铜中毒和其他细胞死亡机制(如铁中毒)之间可能存在复杂的相互作用,但目前仍是一个谜)。在这篇综述中,我们总结了对铜代谢、铜增生症和铜衰竭的分子机制及其潜在的癌症治疗靶点的认识进展。这些新发现推动了以金属化合物为重点的癌症转化研究领域的迅速发展。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
99.40
自引率
0.40%
发文量
114
审稿时长
6-12 weeks
期刊介绍:
Nature Reviews publishes clinical content authored by internationally renowned clinical academics and researchers, catering to readers in the medical sciences at postgraduate levels and beyond. Although targeted at practicing doctors, researchers, and academics within specific specialties, the aim is to ensure accessibility for readers across various medical disciplines. The journal features in-depth Reviews offering authoritative and current information, contextualizing topics within the history and development of a field. Perspectives, News & Views articles, and the Research Highlights section provide topical discussions, opinions, and filtered primary research from diverse medical journals.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


