Molybdenum effects on the stability of passive films unraveled at the nanometer and atomic scales
IF 6.6
2区 材料科学
Q1 MATERIALS SCIENCE, MULTIDISCIPLINARY
引用次数: 0
Abstract
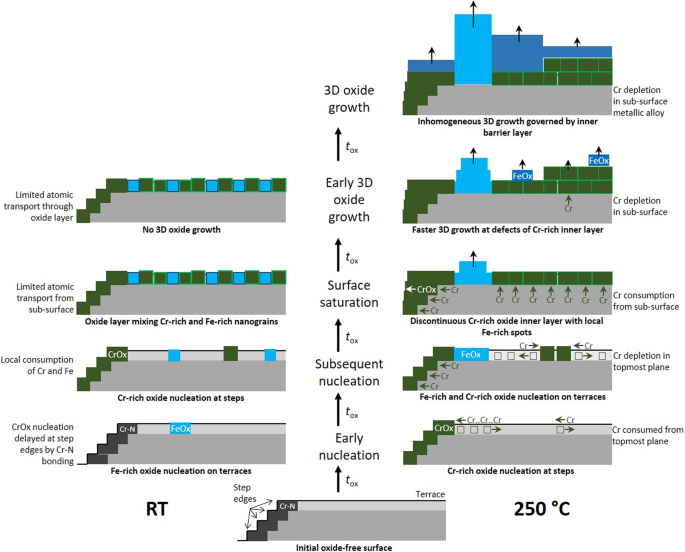
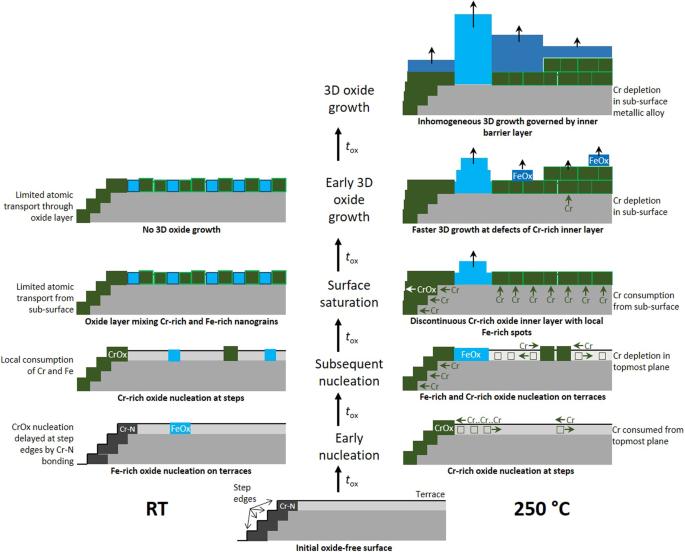
Data recently obtained on model FeCrNi(Mo), 316 L stainless steel, and FeCrNiCo(Mo) passivated surfaces by advanced surface analysis and density functional theory modeling are comprehensively discussed to unravel the multiple effects that molybdenum might have at the nanometer and atomic scales to enhance the stability of passive films. The key role played on corrosion protection by the compositional and structural nanoscale defects of the passive film that originate from the pre-passivation mechanisms of the surface is considered. It is shown how Mo, enriched together with Cr in the nanometer-thick passive film, can combine several effects to enhance the resistance to Cl--induced passivity breakdown. Enriched as Mo(VI) species in the outer exchange layer of the passive film, Mo impedes the deep penetration of Cl- ions and limits their access to the inner barrier layer. Dispersed as Mo(IV) at the interface with the inner layer, Mo protects against the entry of Cl- ions into the defect sites of the Cr(III) oxide barrier. Present as Mo(IV + δ) in the Fe-rich compositional nanoscale defects self-generated by the local failure of Cr supply upon initial formation of the barrier layer, Mo enhances the selective dissolution of iron and its replacement by chromium and molybdenum. By impeding the formation of O vacancies, Mo also increases the resistance against chloride entry in the oxide matrix, thereby curing these the Fe-rich weak sites against Cl--induced passivity breakdown.
在纳米和原子尺度上揭示钼对被动薄膜稳定性的影响
本文全面讨论了最近通过先进的表面分析和密度泛函理论建模获得的铁铬镍(钼)、316 L 不锈钢和铁铬镍钴(钼)钝化模型表面的数据,以揭示钼在纳米和原子尺度上对提高钝化膜稳定性可能产生的多重影响。研究考虑了源于表面预钝化机制的被动膜纳米级成分和结构缺陷对腐蚀防护的关键作用。研究表明,在纳米厚的钝化膜中与铬一起富集的钼可以结合多种效应来增强对 Cl 引起的钝性破坏的抵抗力。作为钼(VI)物种富集在被动膜的外部交换层中,钼阻碍了 Cl- 离子的深度渗透,并限制了它们进入内部阻挡层。钼以 Mo(IV) 的形式分散在与内层的界面上,可防止 Cl- 离子进入铬(III)氧化物阻挡层的缺陷位点。钼以 Mo(IV + δ) 的形式存在于阻挡层最初形成时由于局部铬供应失效而自发产生的富含铁成分的纳米级缺陷中,从而增强了铁的选择性溶解以及铬和钼对铁的替代作用。通过阻碍 O 空位的形成,钼还增加了氧化物基体中氯化物进入的阻力,从而固化了这些富含铁的薄弱位点,使其免受 Cl 引起的钝性破坏。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

npj Materials Degradation
MATERIALS SCIENCE, MULTIDISCIPLINARY-
CiteScore
7.80
自引率
7.80%
发文量
86
审稿时长
6 weeks
期刊介绍:
npj Materials Degradation considers basic and applied research that explores all aspects of the degradation of metallic and non-metallic materials. The journal broadly defines ‘materials degradation’ as a reduction in the ability of a material to perform its task in-service as a result of environmental exposure.
The journal covers a broad range of topics including but not limited to:
-Degradation of metals, glasses, minerals, polymers, ceramics, cements and composites in natural and engineered environments, as a result of various stimuli
-Computational and experimental studies of degradation mechanisms and kinetics
-Characterization of degradation by traditional and emerging techniques
-New approaches and technologies for enhancing resistance to degradation
-Inspection and monitoring techniques for materials in-service, such as sensing technologies
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


