Fluorinated diaminobicyclooxacalixarene and diaminotetraoxacalixarenes: Synthesis and conformational behaviour
IF 1.9
4区 化学
Q3 CHEMISTRY, INORGANIC & NUCLEAR
引用次数: 0
Abstract
Polyfluorinated diaminotetraoxacalixarenes and diaminobicyclooxacalixarene were obtained in good yield by reduction of the corresponding dinitrooxacalixarenes with sodium dithionite. Fluorinated tetraoxacalix[4]arenes with nitro and amino groups located in the inner rim of the macrocycle exists in the solution as an equilibrium mixture of two conformers. For diaminotetraoxacalixarenes the conformational equilibrium is shifted towards conformers having a 1,3-alternate structure. The ESI-MS method shows the possibility of complexing fluorinated diaminotetraoxacalixarenes and diaminobicyclooxacalixarenes with metal cations.
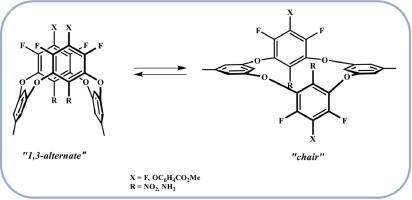
氟化二氨基双环氧杂六烯烃和二氨基四氧杂六烯烃:合成和构象行为
通过用二亚硫酸钠还原相应的二硝基氧杂[4]苊烯,获得了多氟化二氨基四氧杂[4]苊烯和二氨基双环氧杂[4]苊烯,收率很高。氟化四氧杂六[4]烯的硝基和氨基位于大环的内缘,在溶液中以两种构象的平衡混合物形式存在。二氨基四氧杂喹喔啉的构象平衡向具有 1,3 邻位结构的构象转移。ESI-MS 方法显示了氟化二氨基四氧杂六烯烃和二氨基双环氧杂六烯烃与金属阳离子复合的可能性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Fluorine Chemistry
化学-无机化学与核化学
CiteScore
3.80
自引率
10.50%
发文量
99
审稿时长
33 days
期刊介绍:
The Journal of Fluorine Chemistry contains reviews, original papers and short communications. The journal covers all aspects of pure and applied research on the chemistry as well as on the applications of fluorine, and of compounds or materials where fluorine exercises significant effects. This can include all chemistry research areas (inorganic, organic, organometallic, macromolecular and physical chemistry) but also includes papers on biological/biochemical related aspects of Fluorine chemistry as well as medicinal, agrochemical and pharmacological research. The Journal of Fluorine Chemistry also publishes environmental and industrial papers dealing with aspects of Fluorine chemistry on energy and material sciences. Preparative and physico-chemical investigations as well as theoretical, structural and mechanistic aspects are covered. The Journal, however, does not accept work of purely routine nature.
For reviews and special issues on particular topics of fluorine chemistry or from selected symposia, please contact the Regional Editors for further details.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


