IgG exacerbates genital chlamydial pathology in females by enhancing pathogenic CD8 + T cell responses
IF 4.1
4区 医学
Q2 IMMUNOLOGY
引用次数: 0
Abstract
Abstract Chlamydia trachomatis infections are an important sexually transmitted infection that can lead to inflammation, scarring and hydrosalpinx/infertility. However, infections are commonly clinically asymptomatic and do not receive treatment. The underlying cause of asymptomatic immunopathology remains unknown. Here, we demonstrate that IgG produced during male infection enhanced the incidence of immunopathology and infertility in females. Human endocervical cells expressing the neonatal Fc Receptor (FcRn) increased translocation of human IgG‐opsonized C. trachomatis . Using total IgG purified from infected male mice, we opsonized C. muridarum and then infected female mice, mimicking sexual transmission. Following infection, IgG‐opsonized Chlamydia was found to transcytose the epithelial barrier in the uterus, where it was phagocytosed by antigen‐presenting cells (APCs) and trafficked to the draining lymph nodes. APCs then expanded both CD4 + and CD8 + T cell populations and caused significantly more infertility in female mice infected with non‐opsonized Chlamydia . Enhanced phagocytosis of IgG‐opsonized Chlamydia significantly increased pro‐inflammatory signalling and T cell proliferation. As IgG is transcytosed by FcRn, we utilized FcRn −/− mice and observed that shedding kinetics of Chlamydia were only affected in FcRn −/− mice infected with IgG‐opsonized Chlamydia . Depletion of CD8 + T cells in FcRn −/− mice lead to a significant reduction in the incidence of infertility. Taken together, these data demonstrate that IgG seroconversion during male infection can amplify female immunopathology, dependent on FcRn transcytosis, APC differentiation and enhanced CD8 T cell responses.
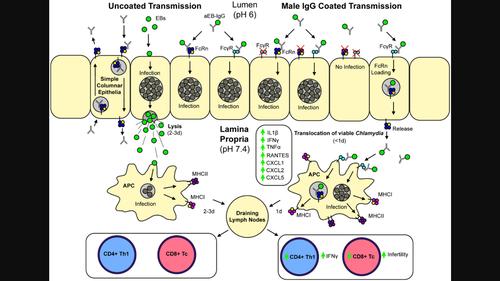
IgG通过增强致病性CD8+ T细胞反应加重女性生殖器衣原体病理
沙眼衣原体感染是一种重要的性传播感染,可导致炎症、瘢痕形成和输卵管积水/不孕。然而,感染通常是临床无症状的,不接受治疗。无症状免疫病理的根本原因尚不清楚。在这里,我们证明在男性感染期间产生的IgG增加了女性免疫病理和不育的发生率。表达新生儿Fc受体(FcRn)的人宫颈内皮细胞增加了人IgG - opsonized沙眼衣原体的易位。利用从感染的雄性小鼠中纯化的总IgG,我们对鼠纹弓形虫进行了免疫,然后感染了雌性小鼠,模拟性传播。在感染后,IgG - opsonized衣原体被发现跨越子宫上皮屏障,在那里它被抗原呈递细胞(apc)吞噬并运输到引流淋巴结。然后APCs扩增CD4 +和CD8 + T细胞群,并在感染非活化衣原体的雌性小鼠中引起更多的不育。IgG -调理衣原体的吞噬作用增强,显著增加促炎信号和T细胞增殖。由于IgG被FcRn转胞,我们利用FcRn - / -小鼠,观察到衣原体的脱落动力学仅在感染了IgG -活化衣原体的FcRn - / -小鼠中受到影响。FcRn - / -小鼠中CD8 + T细胞的耗竭导致不孕症的发生率显著降低。综上所述,这些数据表明,男性感染期间IgG血清转化可以放大女性的免疫病理,依赖于FcRn转胞作用、APC分化和增强的CD8 T细胞反应。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
7.70
自引率
5.40%
发文量
109
审稿时长
1 months
期刊介绍:
This peer-reviewed international journal publishes original articles and reviews on all aspects of basic, translational and clinical immunology. The journal aims to provide high quality service to authors, and high quality articles for readers.
The journal accepts for publication material from investigators all over the world, which makes a significant contribution to basic, translational and clinical immunology.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


