Synthesis of Fluorinated Amines: A Personal Account
IF 3.3
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
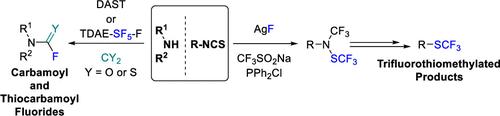
This Account highlights the recent contributions made by our laboratory in the development of novel strategies to synthesize fluorinated amines. These strategies allow the practitioner to efficiently access carbamoyl fluorides, thiocarbamoyl fluorides as well as trifluoromethylamines using CO2 or CS2 as benign C1 sources. In addition, a novel N(SCF3)CF3 moiety was synthesized. Noteworthy, we demonstrated that this reagent could also be used in radical- or electrophilic-based trifluoromethylthiolation reactions.
氟化胺的合成:个人叙述
本报告重点介绍了我们实验室最近在开发合成氟化胺的新策略方面做出的贡献。这些策略使从业人员能够使用二氧化碳或 CS2 作为良性 C1 源,高效地获得氨基甲酰氟、硫代氨基甲酰氟以及三氟甲基胺。此外,我们还合成了一种新型 N(SCF3)CF3 分子。值得注意的是,我们证明这种试剂还可用于基于自由基或亲电的三氟甲基硫代反应。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Organic & Inorganic Au
有机化学、无机化学-
CiteScore
4.10
自引率
0.00%
发文量
0
期刊介绍:
ACS Organic & Inorganic Au is an open access journal that publishes original experimental and theoretical/computational studies on organic organometallic inorganic crystal growth and engineering and organic process chemistry. Short letters comprehensive articles reviews and perspectives are welcome on topics that include:Organic chemistry Organometallic chemistry Inorganic Chemistry and Organic Process Chemistry.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


