Abnormal myosin post-translational modifications and ATP turnover time associated with human congenital myopathy-related RYR1 mutations
IF 5.6
2区 医学
Q1 PHYSIOLOGY
引用次数: 1
Abstract
Conditions related to mutations in the gene encoding the skeletal muscle ryanodine receptor 1 (RYR1) are genetic muscle disorders and include congenital myopathies with permanent weakness, as well as episodic phenotypes such as rhabdomyolysis/myalgia. Although RYR1 dysfunction is the primary mechanism in RYR1‐related disorders, other downstream pathogenic events are less well understood and may include a secondary remodeling of major contractile proteins. Hence, in the present study, we aimed to investigate whether congenital myopathy‐related RYR1 mutations alter the regulation of the most abundant contractile protein, myosin.
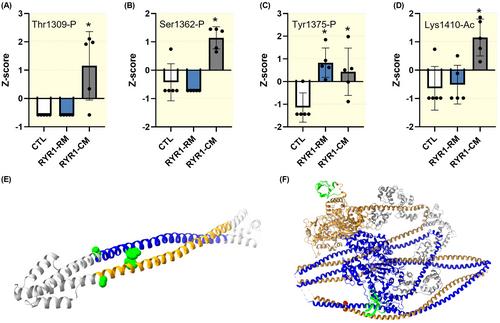
与人类先天性肌病相关RYR1突变相关的异常肌球蛋白翻译后修饰和ATP周转时间。
目的:与编码骨骼肌ryanodine受体1(RYR1)基因突变相关的疾病是遗传性肌肉疾病,包括先天性肌病伴永久性无力,以及偶发性表型,如横纹肌溶解症/肌痛。尽管RYR1功能障碍是RYR1相关疾病的主要机制,但其他下游致病事件尚不清楚,可能包括主要收缩蛋白的二次重塑。因此,在本研究中,我们旨在研究先天性肌病相关的RYR1突变是否会改变最丰富的收缩蛋白肌球蛋白的调节。方法:我们使用了5名RYR1相关先天性肌病患者的骨骼肌组织,并将其与5名对照组和5名RYR1-相关横纹肌溶解症/肌痛患者进行了比较。然后,我们使用LC/MS定义了肌球蛋白重链(MyHC)的翻译后修饰。同时,我们使用Mant-ATP追逐实验确定了肌球蛋白弛豫状态,并进行了分子动力学(MD)模拟。结果:LC/MS显示先天性肌病患者的β/慢MyHC上有两个额外的磷酸化(Thr1309-P和Ser1362-P)和一个乙酰化(Lys1410-Ac)。该方法还鉴定了这些患者的MyHC IIa型上缺乏的六种乙酰化(Lys35-Ac、Lys663-Ac、Lys763-Ac、LYs11171Ac、Lys1360-Ac和Lys1733-Ac)。MD模拟表明,修饰肌球蛋白Ser1362影响蛋白质结构和动力学。最后,Mant-ATP追逐实验表明,在无序的松弛构象中,肌球蛋白头的ATP周转时间更快。结论:总之,我们的研究结果表明,RYR1突变对肌球蛋白结构和功能具有次要的负面影响,可能导致先天性肌病表型。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Acta Physiologica
医学-生理学
CiteScore
11.80
自引率
15.90%
发文量
182
审稿时长
4-8 weeks
期刊介绍:
Acta Physiologica is an important forum for the publication of high quality original research in physiology and related areas by authors from all over the world. Acta Physiologica is a leading journal in human/translational physiology while promoting all aspects of the science of physiology. The journal publishes full length original articles on important new observations as well as reviews and commentaries.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


