改造环状 RNA 以提高蛋白质产量
IF 33.1
1区 生物学
Q1 BIOTECHNOLOGY & APPLIED MICROBIOLOGY
引用次数: 54
摘要
环状 RNA(circRNA)是真核细胞中稳定而普遍的 RNA,由反向剪接产生。合成的 circRNA 和一些内源性 circRNA 可以编码蛋白质,这使 circRNA 有希望成为基因表达的平台。在这项研究中,我们开发了一种系统化的方法,用于快速组装和测试影响合成 circRNA 蛋白生成的特征。为了最大限度地提高 circRNA 的翻译效率,我们优化了五个要素:载体拓扑结构、5′和 3′非翻译区、内部核糖体进入位点以及招募翻译启动机制的合成适配体。这些设计原则合在一起,可使环状 RNA 蛋白产量提高数百倍,体外翻译量超过信使 RNA,体内翻译更持久,并可在多个转基因中通用。通过优化载体设计,环状 RNA 的蛋白质表达量提高了几百倍。本文章由计算机程序翻译,如有差异,请以英文原文为准。
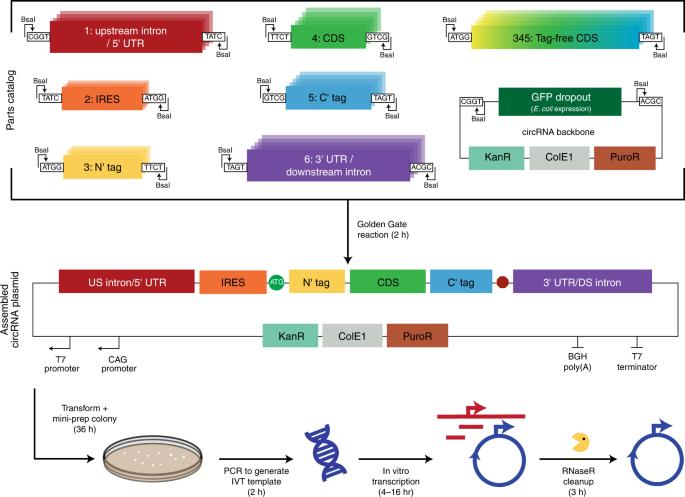
Engineering circular RNA for enhanced protein production
Circular RNAs (circRNAs) are stable and prevalent RNAs in eukaryotic cells that arise from back-splicing. Synthetic circRNAs and some endogenous circRNAs can encode proteins, raising the promise of circRNA as a platform for gene expression. In this study, we developed a systematic approach for rapid assembly and testing of features that affect protein production from synthetic circRNAs. To maximize circRNA translation, we optimized five elements: vector topology, 5′ and 3′ untranslated regions, internal ribosome entry sites and synthetic aptamers recruiting translation initiation machinery. Together, these design principles improve circRNA protein yields by several hundred-fold, provide increased translation over messenger RNA in vitro, provide more durable translation in vivo and are generalizable across multiple transgenes. Protein expression from circular RNAs is enhanced several hundred-fold by optimizing vector design.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Nature biotechnology
工程技术-生物工程与应用微生物
CiteScore
63.00
自引率
1.70%
发文量
382
审稿时长
3 months
期刊介绍:
Nature Biotechnology is a monthly journal that focuses on the science and business of biotechnology. It covers a wide range of topics including technology/methodology advancements in the biological, biomedical, agricultural, and environmental sciences. The journal also explores the commercial, political, ethical, legal, and societal aspects of this research.
The journal serves researchers by providing peer-reviewed research papers in the field of biotechnology. It also serves the business community by delivering news about research developments. This approach ensures that both the scientific and business communities are well-informed and able to stay up-to-date on the latest advancements and opportunities in the field.
Some key areas of interest in which the journal actively seeks research papers include molecular engineering of nucleic acids and proteins, molecular therapy, large-scale biology, computational biology, regenerative medicine, imaging technology, analytical biotechnology, applied immunology, food and agricultural biotechnology, and environmental biotechnology.
In summary, Nature Biotechnology is a comprehensive journal that covers both the scientific and business aspects of biotechnology. It strives to provide researchers with valuable research papers and news while also delivering important scientific advancements to the business community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


