作为“手性氨等价物”的O-(α-苯乙基)羟胺:5-氧吡咯烷和6-氧吡啶-3羧酸的合成和拆分
Q2 Chemistry
引用次数: 3
摘要
描述了一种合成和分离五元和六元内酰胺(即5-氧吡咯烷和6-氧吡啶-3-羧酸)的方法。该方法依赖于一锅迈克尔反应-衣康酸或高衣康酸二乙基与对映纯O-(α-苯乙基)羟胺的环化,作为“手性氨等价物”。结果表明,该手性助剂可用于内酰胺非对映异构体产物的分离,并易于催化氢解去除。本文章由计算机程序翻译,如有差异,请以英文原文为准。
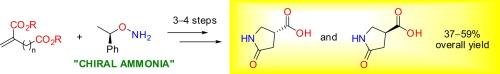
O-(α-Phenylethyl)hydroxylamine as a ‘chiral ammonia equivalent’: synthesis and resolution of 5-oxopyrrolidine- and 6-oxopiperidine-3-carboxylic acids
An approach to the synthesis and resolution of five- and six-membered lactams (i.e., 5-oxopyrrolidine- and 6-oxopiperidine-3-carboxylic acids) is described. The method relies on the one-pot Michael reaction—cyclization of itaconic acid or diethyl homoitaconate and enantiopure O-(α-phenylethyl)hydroxylamine as a ‘chiral ammonia equivalent’. It is shown that this chiral auxiliary can be used for the separation of diastereomeric lactam products and then easily removed by catalytic hydrogenolysis.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Tetrahedron, asymmetry
化学-无机化学与核化学
CiteScore
4.70
自引率
0.00%
发文量
0
审稿时长
1 months
期刊介绍:
Cessation. Tetrahedron: Asymmetry presents experimental or theoretical research results of outstanding significance and timeliness on asymmetry in organic, inorganic, organometallic and physical chemistry, as well as its application to related disciplines, especially bio-organic chemistry.
The journal publishes critical reviews, original research articles and preliminary communications dealing with all aspects of the chemical, physical and theoretical properties of non-racemic organic and inorganic materials and processes. Topics relevant to the journal include: the physico-chemical and biological properties of enantiomers; strategies and methodologies of asymmetric synthesis; resolution; chirality recognition and enhancement; analytical techniques for assessing enantiomeric purity and the unambiguous determination of absolute configuration; and molecular graphics and modelling methods for interpreting and predicting asymmetric phenomena. Papers describing the synthesis or properties of non-racemic molecules will be required to include a separate statement in the form of a Stereochemistry Abstract, for publication in the same issue, of the criteria used for the assignment of configuration and enantiomeric purity.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


