立体选择性多米诺环化/还原反应合成多羟基化双环四氢呋喃和四氢吡喃
Q2 Chemistry
引用次数: 1
摘要
描述了一种新的反应级联,涉及路易斯酸诱导的异丙基保护基团的迁移,随后形成吡喃糖或呋喃糖环,随后半缩醛的还原。根据反应条件以及底物的立体化学性质,可以以合理的产率得到多羟基化的四氢呋喃或四氢吡喃。该转化中使用的合成物是通过高度立体选择性的一锅串联反应制备的,包括乙烯基溴化镁与d-葡萄糖衍生的环己酮的1,4- michael加成,然后醛醇与2,3- o -异丙基-d-甘油醛的反应。本文章由计算机程序翻译,如有差异,请以英文原文为准。
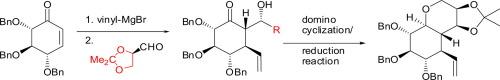
Synthesis of polyhydroxylated bicyclic tetrahydrofurans and tetrahydropyrans via a stereoselective domino cyclization/reduction reaction
A novel reaction cascade involving a Lewis acid-induced migration of an isopropylidene protecting group followed by the formation of a pyranose or furanose ring and subsequent reduction of the hemiacetal is described. Depending on the reaction conditions, as well as, the stereochemistry of the substrate, polyhydroxylated tetrahydrofurans or tetrahydropyrans can be obtained in reasonable yields. The synthons used in this transformation were prepared via a highly stereoselective one-pot tandem reaction, consisting of a 1,4-Michael addition of vinylmagnesium bromide to d-glucose-derived cyclohexenone followed by aldol reaction with 2,3-O-isopropylidene-d-glyceraldehyde.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Tetrahedron, asymmetry
化学-无机化学与核化学
CiteScore
4.70
自引率
0.00%
发文量
0
审稿时长
1 months
期刊介绍:
Cessation. Tetrahedron: Asymmetry presents experimental or theoretical research results of outstanding significance and timeliness on asymmetry in organic, inorganic, organometallic and physical chemistry, as well as its application to related disciplines, especially bio-organic chemistry.
The journal publishes critical reviews, original research articles and preliminary communications dealing with all aspects of the chemical, physical and theoretical properties of non-racemic organic and inorganic materials and processes. Topics relevant to the journal include: the physico-chemical and biological properties of enantiomers; strategies and methodologies of asymmetric synthesis; resolution; chirality recognition and enhancement; analytical techniques for assessing enantiomeric purity and the unambiguous determination of absolute configuration; and molecular graphics and modelling methods for interpreting and predicting asymmetric phenomena. Papers describing the synthesis or properties of non-racemic molecules will be required to include a separate statement in the form of a Stereochemistry Abstract, for publication in the same issue, of the criteria used for the assignment of configuration and enantiomeric purity.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


