拟南芥基因体甲基化处于选择过程中。
IF 3.3
3区 生物学
引用次数: 13
摘要
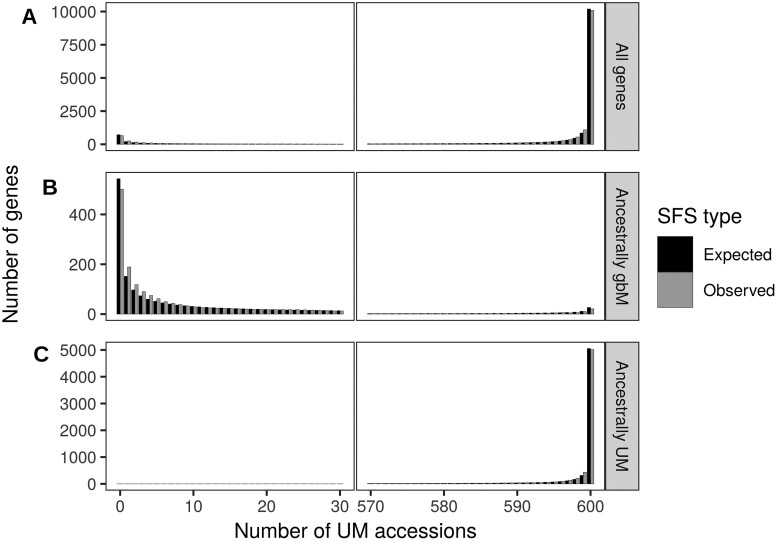
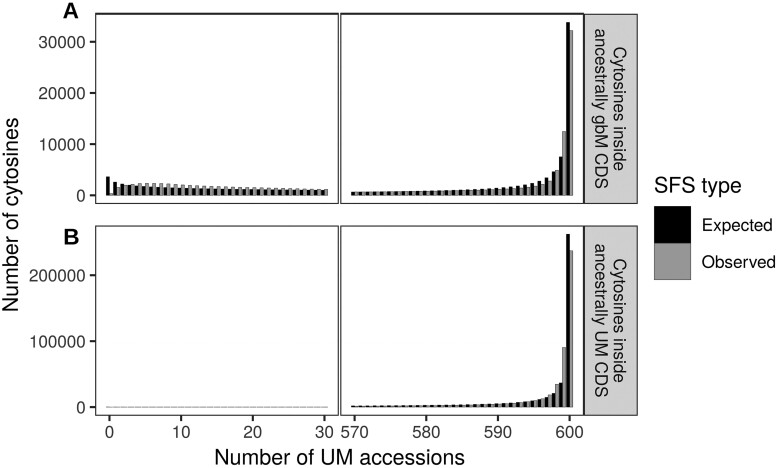
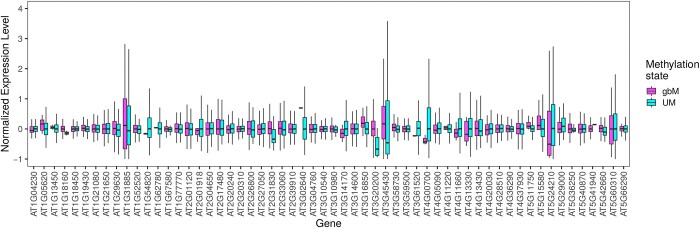
在植物、哺乳动物和昆虫中,一些基因在CG二核苷酸环境下甲基化,这种现象被称为基因体甲基化(gbM)。这一现象是否有任何功能作用一直存在争议。本研究利用拟南芥876个叶片甲基化组的可用性,在基因水平上表征了甲基化的群体频率,并估计了等位基因状态的位点频谱。使用专门为表观遗传数据设计的群体遗传学模型,我们发现具有祖先gbM的基因在显着选择下保持甲基化。相反,祖先未甲基化的基因在选择下保持未甲基化。在单个胞嘧啶水平上重复分析证实了这些结果。估计的选择系数很小,约为4 Nes = 1.4,这与选择对密码子使用的影响程度相似。我们还估计拟南芥失去gbM的速度比获得gbM的速度快三倍,这可能是由于最近转向自交后选择效率的降低。最后,我们通过与基因表达的联系研究了gbM的潜在功能。在具有多态性甲基化状态的基因中,基因体甲基化等位基因的表达一致且显著高于非甲基化等位基因。尽管很难将遗传效应与表观遗传效应区分开来,但我们的研究表明,gbM对适应度有一个小但可测量的影响,这可能是由于它与表型样基因表达有关。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Gene body methylation is under selection in Arabidopsis thaliana.
Abstract In plants, mammals and insects, some genes are methylated in the CG dinucleotide context, a phenomenon called gene body methylation (gbM). It has been controversial whether this phenomenon has any functional role. Here, we took advantage of the availability of 876 leaf methylomes in Arabidopsis thaliana to characterize the population frequency of methylation at the gene level and to estimate the site-frequency spectrum of allelic states. Using a population genetics model specifically designed for epigenetic data, we found that genes with ancestral gbM are under significant selection to remain methylated. Conversely, ancestrally unmethylated genes were under selection to remain unmethylated. Repeating the analyses at the level of individual cytosines confirmed these results. Estimated selection coefficients were small, on the order of 4 Nes = 1.4, which is similar to the magnitude of selection acting on codon usage. We also estimated that A. thaliana is losing gbM threefold more rapidly than gaining it, which could be due to a recent reduction in the efficacy of selection after a switch to selfing. Finally, we investigated the potential function of gbM through its link with gene expression. Across genes with polymorphic methylation states, the expression of gene body methylated alleles was consistently and significantly higher than unmethylated alleles. Although it is difficult to disentangle genetic from epigenetic effects, our work suggests that gbM has a small but measurable effect on fitness, perhaps due to its association to a phenotype-like gene expression.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Genetics
生物-遗传学
CiteScore
6.20
自引率
6.10%
发文量
177
期刊介绍:
GENETICS is published by the Genetics Society of America, a scholarly society that seeks to deepen our understanding of the living world by advancing our understanding of genetics. Since 1916, GENETICS has published high-quality, original research presenting novel findings bearing on genetics and genomics. The journal publishes empirical studies of organisms ranging from microbes to humans, as well as theoretical work.
While it has an illustrious history, GENETICS has changed along with the communities it serves: it is not your mentor''s journal.
The editors make decisions quickly – in around 30 days – without sacrificing the excellence and scholarship for which the journal has long been known. GENETICS is a peer reviewed, peer-edited journal, with an international reach and increasing visibility and impact. All editorial decisions are made through collaboration of at least two editors who are practicing scientists.
GENETICS is constantly innovating: expanded types of content include Reviews, Commentary (current issues of interest to geneticists), Perspectives (historical), Primers (to introduce primary literature into the classroom), Toolbox Reviews, plus YeastBook, FlyBook, and WormBook (coming spring 2016). For particularly time-sensitive results, we publish Communications. As part of our mission to serve our communities, we''ve published thematic collections, including Genomic Selection, Multiparental Populations, Mouse Collaborative Cross, and the Genetics of Sex.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


