CO-PERK-IRG1轴减轻肝缺血再灌注损伤中的氧化应激和铁下垂
IF 8.2
2区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
肝缺血再灌注损伤(IRI)可作为外科手术的结果,包括肝移植。肝脏IRI的特征是氧化应激、炎症和激活多种受调节的细胞死亡途径(RCD)。在RCD中,铁下垂是由增强的脂质过氧化和活性氧(ROS)形成驱动的。一氧化碳(CO)具有细胞保护和抗炎特性,在各种临床前模型中,当低浓度应用时,可以减轻IRI,尽管潜在的机制尚不清楚。在这项研究中,我们描述了蛋白激酶rna样内质网激酶(PERK)信号在CO对氧化应激和铁凋亡的保护作用中的关键作用。我们证明,co诱导的PERK激活使细胞抗氧化反应的主要调节因子Nrf2磷酸化,导致KCs和肝细胞中免疫应答基因1 (IRG1)表达上调。irg1衍生的衣康酸通过提高GPX4水平抑制ROS积累和铁下垂,同时降低ACSL4和PTGS2的表达。使用siRNA或敲除小鼠对PERK或Nrf2进行遗传干扰,可消除co介导的IRG1诱导和对铁凋亡的保护作用,突出了该途径不可或缺的作用。此外,在肝脏IRI的体内模型中,CO治疗以依赖于PERK-Nrf2-IRG1轴的方式显著减少炎症、组织病理学损伤和铁上吊。这些发现确立了perk - nrf2 - irg1 -衣康酸途径作为肝脏IRI和其他可能由铁中毒引起的疾病的新治疗靶点。本文章由计算机程序翻译,如有差异,请以英文原文为准。
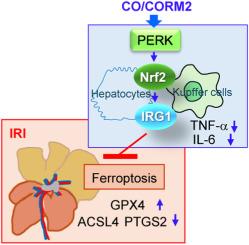
CO-PERK-IRG1 axis attenuates oxidative stress and ferroptosis in hepatic ischemia-reperfusion injury
Hepatic ischemia-reperfusion injury (IRI) can arise as the consequence of surgical procedures, including liver transplant. Hepatic IRI is characterized by oxidative stress, inflammation, and activation of multiple regulated cell death pathways (RCD). Among RCD, ferroptosis is driven by enhanced lipid peroxidation and reactive oxygen species (ROS) formation. Carbon monoxide (CO), which can exert cytoprotective and anti-inflammatory properties, can mitigate IRI in various pre-clinical models when applied at low concentration, though the underlying mechanisms remain poorly understood. In this study, we describe a critical role of protein kinase RNA-like endoplasmic reticulum kinase (PERK) signaling in the protective effects of CO against oxidative stress and ferroptosis. We demonstrate that CO-induced PERK activation phosphorylates Nrf2, a master regulator of the cellular antioxidant response, leading to upregulation of immune-responsive gene 1 (IRG1) expression in both Kupffer cells (KCs) and hepatocytes. IRG1-derived itaconate production suppressed ROS accumulation and ferroptosis by enhancing GPX4 levels, while reducing ACSL4 and PTGS2 expression. Genetic interference of PERK or Nrf2 using siRNA or knockout mice abolished CO-mediated IRG1 induction and protection against ferroptosis, highlighting the indispensable role of this pathway.
Furthermore, in an in vivo model of hepatic IRI, CO treatment significantly reduced inflammation, histopathological damage, and ferroptosis in a manner dependent on the PERK-Nrf2-IRG1 axis. These findings establish the PERK-Nrf2-IRG1-itaconate pathway as a novel therapeutic target for hepatic IRI and potentially other ferroptosis-driven conditions.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Free Radical Biology and Medicine
医学-内分泌学与代谢
CiteScore
14.00
自引率
4.10%
发文量
850
审稿时长
22 days
期刊介绍:
Free Radical Biology and Medicine is a leading journal in the field of redox biology, which is the study of the role of reactive oxygen species (ROS) and other oxidizing agents in biological systems. The journal serves as a premier forum for publishing innovative and groundbreaking research that explores the redox biology of health and disease, covering a wide range of topics and disciplines. Free Radical Biology and Medicine also commissions Special Issues that highlight recent advances in both basic and clinical research, with a particular emphasis on the mechanisms underlying altered metabolism and redox signaling. These Special Issues aim to provide a focused platform for the latest research in the field, fostering collaboration and knowledge exchange among researchers and clinicians.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


